The shift from “pharmacy of the world” to a global innovator hub is being led by a select group of companies that have moved beyond basic manufacturing to embrace high-stakes clinical R&D and global commercial infrastructure. Here are the 10 Indian pharmaceutical and Medtech leaders poised for global dominance.
1. Sun Pharmaceutical Industries: The Specialty Giant
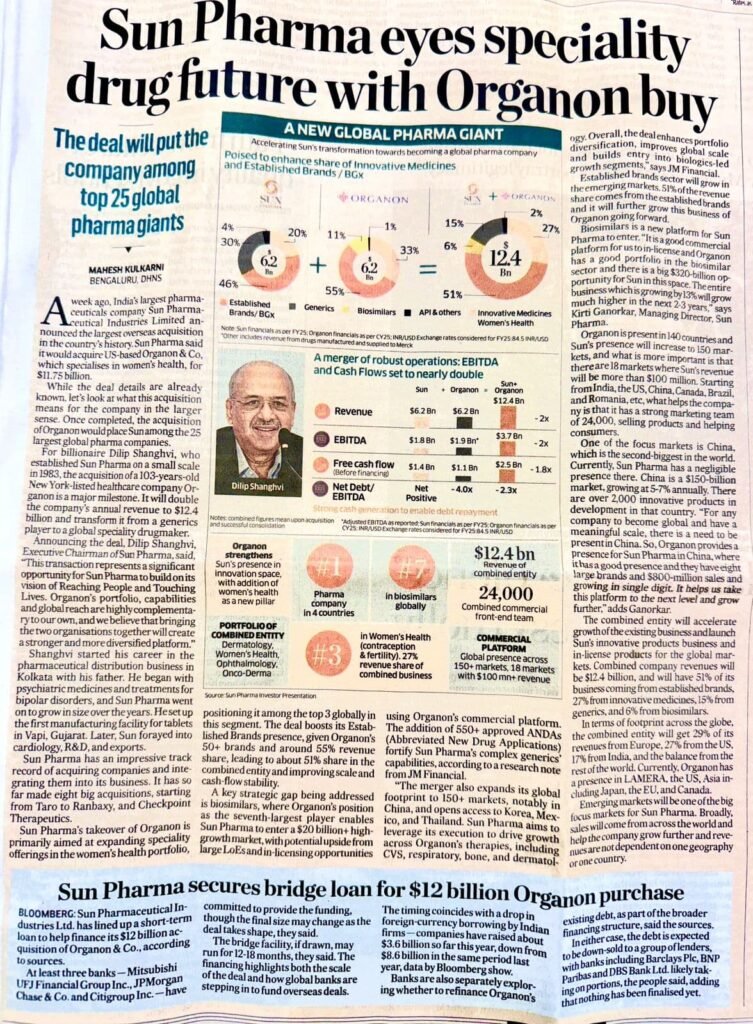
- Strategic Pivot: Transforming from a generic player to a specialty powerhouse through the $11.75 billion acquisition of Organon & Co.
- Global Footprint: Gains an immediate #3 global revenue share in Women’s Health (contraception and fertility) and a top 10 position in global biosimilars.
- Scale: Operates a combined commercial team of 24,000 members across 150+ markets, significantly expanding its reach in China.
2. Meril Life Sciences: The Vascular Innovator
- Core Strength: Specialized in high-precision cardiovascular intervention and structural heart technology.
- Technological Moat: Developed the *Myval THV, the first Indian TAVR system to receive a CE mark, and the *BioMime DES, which features ultra-thin 65-micron struts for better clinical outcomes.
- Market Strategy: Competes with global giants by establishing direct subsidiaries in the US and Europe to manage physician relationships and complex clinical trials.
3. Dr. Reddy’s Laboratories: The Peptide Pioneer
- Innovation Focus: Leading the transition into metabolic health and complex biologics.
- Recent Success: Secured the first generic approval for Semaglutide (GLP-1) in Canada in early 2026, supported by their vertically integrated “OneSource” manufacturing platform.
- Growth Engine: Shifting R&D toward high-barrier “horizon 2 and 3” products, including gene therapy and digital therapeutics.
4. Cipla: The Respiratory Leader
- Strategic Niche: Pursuing “Lung Leadership” through advanced inhalation and drug-device combinations.
- Leadership Change: Appointed Achin Gupta as CEO in April 2026 to lead a pivot toward “next-gen” innovation and digital-led growth.
- Pipeline: Actively launching complex respiratory assets (like generic Advair) and GLP-1 peptides to diversify from traditional primary care.
5. Biocon Biologics: The Biosimilar Specialist
- Portfolio Shift: A pure-play biologics challenger with 60% of total revenue now derived from biosimilars.
- Global Consolidation: Raised $460 million in early 2026 to complete the acquisition of Viatris’s global biosimilars business, granting them full control of their international commercial footprint.
- Impact: Positioned as a key player in reducing the cost of life-saving biologics in the US and Europe.
6. Zydus Lifesciences: The NCE Challenger
- Specialty Pivot: Moving into New Chemical Entities (NCEs) and Orphan Drugs.
- Key Milestone: Received USFDA approval for Zycubo in January 2026, targeting Menkes disease—a significant step into the ultra-rare therapeutic space.
- Diversification: Using targeted M&A to build a specialized portfolio in liver health and wellness.
7. Healthium Medtech: The Surgical Expert
- Core Business: Global leader in surgical sutures, needles, and advanced wound care.
- Competitive Edge: One of the few companies globally with an integrated manufacturing chain for high-end surgical consumables, serving over 90 countries.
- Scaling Strategy: Transitioning from an OEM partner to a branded global player with a focus on surgical innovation.
8. Poly Medicure (Polymed): The Device Designer
- Sector Focus: Infusion therapy, dialysis, and oncology delivery systems.
- R&D Strength: Holds over 160 patents and exports to 110+ countries, shifting from “made in India” to “designed in India” for complex clinical environments.
- Expansion: Actively building capacity for specialized dialysis equipment to meet global chronic care demands.
9. Transasia Bio-Medicals: The Diagnostic Disruptor
- Clinical Focus: In-vitro diagnostics (IVD) including biochemistry, hematology, and immunology.
- Growth Model: Utilizing international acquisitions to gain R&D centers in Europe and the US, enabling them to offer high-end diagnostic tools at an emerging-market price point.
- Reach: Currently India’s largest IVD player with an expanding footprint in 100+ countries.
10. Trivitron Healthcare: The Integrated Medtech Hub
- Niche Markets: Neonatal screening, medical imaging, and lab diagnostics.
- Global Strategy: Employs a unique “Innovation + Manufacturing” hub model, producing specialized equipment in India, Turkey, and Finland for a global audience.
- Future Pivot: Moving heavily into molecular diagnostics and AI-driven imaging solutions.

Appendix: Reference Library
1. Transactional & Financial Data (The Sun-Organon Deal)
- Sun Pharma Investor Relations: The primary source for the $11.75 billion valuation, the $12 billion bridge loan details, and the projected $12.4 billion combined revenue.
- Organon & Co. SEC Filings: Provides the “mirror” data for the deal, confirming their #3 global position in Women’s Health and their substantial infrastructure in the Chinese market.
- Bloomberg Terminal / Financial News: Validates the consortium of lenders (Mitsubishi UFJ, JPMorgan, Citigroup) and the 12-18 month timeline for the bridge facility.
2. Regulatory & Clinical Proof Points
- US Food & Drug Administration (USFDA): Essential for verifying the approval of “Innovator” products from Indian firms, such as Zydus Lifesciences’ Zycubo (January 2026) for Menkes disease.
- Health Canada (Drug Product Database): Confirms Dr. Reddy’s Laboratories’ landmark generic Semaglutide approval in April 2026, a key marker for the “Pharma Renaissance.”
- European Medicines Agency (EMA): Reference for Meril Life Sciences’ CE Mark certifications for the Myval THV (TAVR system) and BioMime stents, proving their global clinical parity.
3. Market Intelligence & Industry Analysis
- IQVIA (formerly IMS Health): For global market share data in specialty therapeutic areas like Biosimilars (where the Sun-Organon entity ranks #7) and Respiratory (Cipla’s core focus).
- JM Financial / Jefferies Research: Specific institutional research notes (e.g., JM Financial’s analysis mentioned in the provided data) that detail the impact of 550+ approved ANDAs on Sun Pharma’s complex generic capabilities.
- The Pharmaletter / Scrip: Trade journals that track the strategic pivots of the “Top 10” mentioned, particularly the leadership transitions and M&A activity of Cipla and Biocon.
4. Medtech Performance Standards
- Medtech Europe / AdvaMed: Benchmarks for the global Medtech expansion of players like Healthium and Polymed, specifically regarding R&D spend and patent filings.
- ClinicalTrials.gov: To reference specific trial results, such as Meril’s TALENT trial, which provides the scientific “moat” required to challenge incumbents in the vascular intervention space.









