Hot topics
Why Most AI Healthcare Tools Stall at the Pilot Stage
Artificial intelligence (AI) in healthcare consistently demonstrates remarkable technical...
Dr. Peter Velev, CEO of CredoWeb discussing Digital in Pharma with MedicinMan
The digital revolution is the fourth industrial revolution. It changes models, processes and whole public sectors. In many reports you will find that, in terms of digitalization, pharma is just next to the public sector, as least digitalized. Pharma just started late. There are many reasons for that – it is one of the most regulated industries with lots of sensitive data involved and many ethical aspects. But the slowed down digitalization has a lot to do with some subjective reasons. Adopting digital technologies requires changing existing models which requires a different mindset – and this is difficult to achieve. Nowadays, in the situation of a global pandemic, we see that digital communication is not only necessary to be successful but it is a must in order to adapt to the New Normality.
Talent Management
A diverse, summer issue with articles on topics ranging from content marketing to the role of experience in a VUCA world. Also, the latest book for Medical Reps reviewed by Prof. Vivek Hattangadi
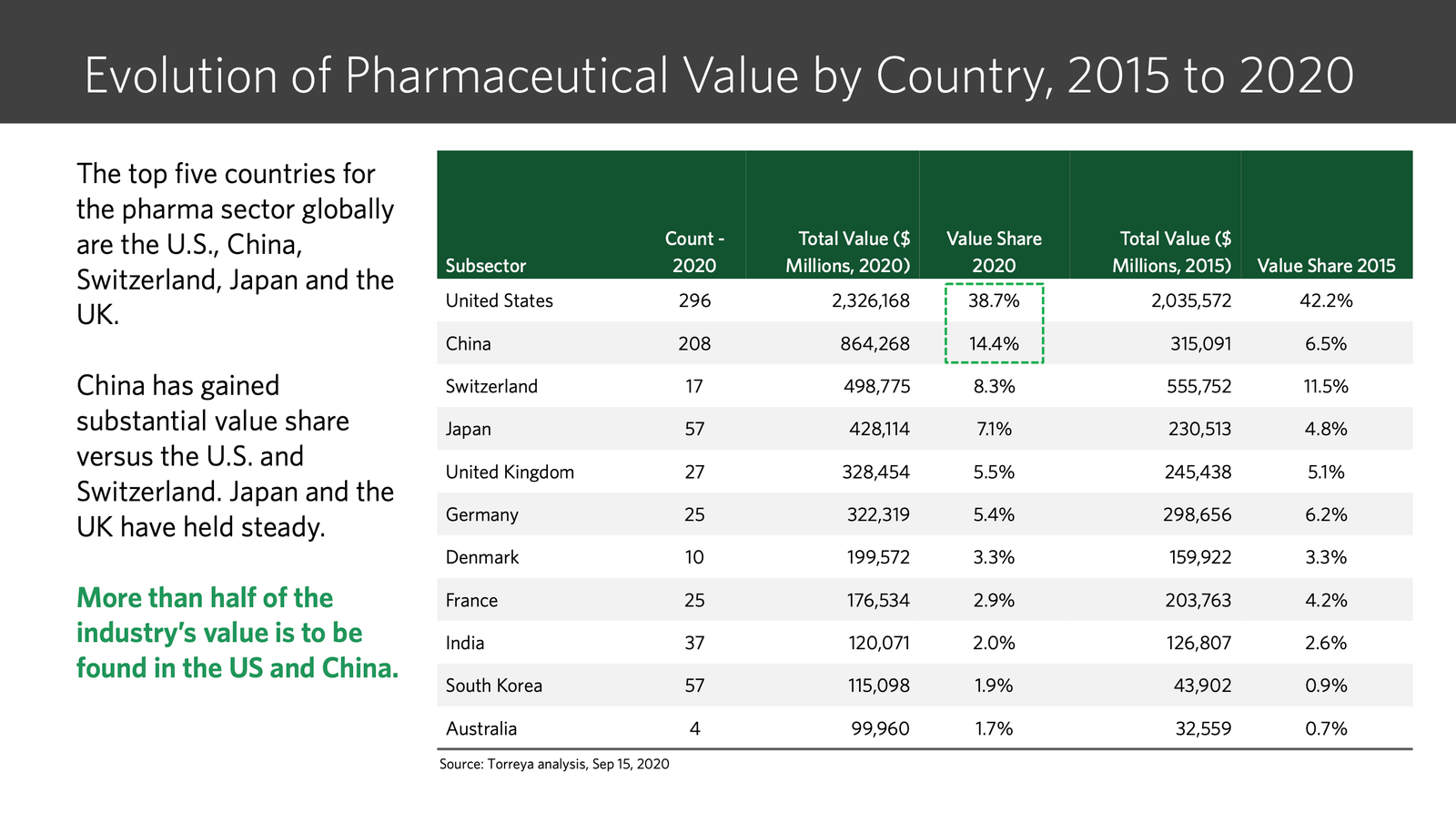
India’s Patent Revocations: Strengthening Access and Challenging Evergreening
The Indian Patent Office’s recent decision to revoke Novartis’...
Cipla’s Leadership Transition from Umang Vohra to Achin Gupta: Succession Planning in Action
By Anup Soans | MedicinMan
Cipla Limited has announced a...
Do Retail Pharmacies Really Care about Patients?
The chemist shop—jammed between a chai stall and a...
Indian Oncology: An Unasked Question, the Challenges Ahead
A Brief History of Scale and an Unasked Question
In...
Are Quick Remedies Keeping Patients Away from Doctors?
The recent Mint Lounge analysis captured a striking consumer...
From Tragedy to Trust: What the CDCSO Can Learn from Dr. Frances Oldham Kelsey
Abstract
The U.S. Food and Drug Administration (FDA) evolved from...
Biocon’s Landmark Succession Planning: Claire Mazumdar is the Future Leader
In a landmark decision for India’s biotechnology sector, Kiran...
Do Retail Pharmacies Really Care about Patients?
The chemist shop—jammed between a chai stall and a...
Indian Oncology: An Unasked Question, the Challenges Ahead
A Brief History of Scale and an Unasked Question
In...
Are Quick Remedies Keeping Patients Away from Doctors?
The recent Mint Lounge analysis captured a striking consumer...
From Tragedy to Trust: What the CDCSO Can Learn from Dr. Frances Oldham Kelsey
Abstract
The U.S. Food and Drug Administration (FDA) evolved from...
Biocon’s Landmark Succession Planning: Claire Mazumdar is the Future Leader
In a landmark decision for India’s biotechnology sector, Kiran...
Do Retail Pharmacies Really Care about Patients?
The chemist shop—jammed between a chai stall and a...
Indian Oncology: An Unasked Question, the Challenges Ahead
A Brief History of Scale and an Unasked Question
In...
Are Quick Remedies Keeping Patients Away from Doctors?
The recent Mint Lounge analysis captured a striking consumer...
From Tragedy to Trust: What the CDCSO Can Learn from Dr. Frances Oldham Kelsey
Abstract
The U.S. Food and Drug Administration (FDA) evolved from...
Biocon’s Landmark Succession Planning: Claire Mazumdar is the Future Leader
In a landmark decision for India’s biotechnology sector, Kiran...
Do Retail Pharmacies Really Care about Patients?
The chemist shop—jammed between a chai stall and a...
Indian Oncology: An Unasked Question, the Challenges Ahead
A Brief History of Scale and an Unasked Question
In...
Are Quick Remedies Keeping Patients Away from Doctors?
The recent Mint Lounge analysis captured a striking consumer...
From Tragedy to Trust: What the CDCSO Can Learn from Dr. Frances Oldham Kelsey
Abstract
The U.S. Food and Drug Administration (FDA) evolved from...
Biocon’s Landmark Succession Planning: Claire Mazumdar is the Future Leader
In a landmark decision for India’s biotechnology sector, Kiran...
Hot topics
Digital Darwinism or Digital Expert – The Choice for Indian Pharma is Clear
It is heartening to see that Pharma marketers have...
Torrent Pharma–JB Chemicals Acquisition Report
Summary
Torrent Pharmaceuticals (TRP) has signed definitive agreements...
How to Harness the Power of Omnichannel Marketing in the Pharma Industry
Pharmaceutical & medical devices Industry is fast adapting to these changes at various levels of the value chain. Billions of dollars are being spent on digital transformation projects ranging from usage of AI & ML in product development, production and predictive maintenance, Blockchain in supply chain and data management & analytics in strategic decision making.
This article focuses on Omnichannel marketing, one of the key digital transformations that the industry is learning and implementing at a rapid pace.
Omnichannel Marketing: Promise and Possibilities
An industry that historically focused all their brand building and market shaping efforts through F2F meetings & medical education programs has started making giant strides in multi-channel strategy now.
A digital journey that started with an e-detailing platform has progressed fast by adding brand websites, Approved E-Mails, social media, webinars and other media effectively creating a multi-channel strategy. The only hindside to this is, all these channels work independently, expecting HCPs to find and sort out information themselves.
Inspirational Frontline Leadership
The last issue of 2016, with articles from K. Hariram and Vivek Hattangadi and new authors, Dr. Ashwin Bonde and Diksha Fouzdar. Diksha's article is the highlight of this issue - an in-depth look at how to embed compliance in the very DNA of a pharma company.
Medical Rep to Chairman & Managing Director – Sunil Attavar’s Success Story
Q 1. Tell us about your journey as a pharma entrepreneur and what made you venture into pharma and stay on in pharma?
My journey started with two good decisions, one- to be a Medical Representative (MR) in Mumbai, which built a strong foundation and the other, to join Helios, a new pharma company, which was like a baptism of fire. Together, they molded me well.
I was promoted as a Front Line Manager (FLM) within a year. And after spending four excellent years in Helios, I joined Group Pharma as Product Manager (PM) and ever since, it’s been an exciting learning experience.
I was exposed to Pharma very early in life as my dad was in J L Morison, as part of the promoter-team of Warren Pharma. At home, I was the designated telephone operator and order processing clerk. Those were the days of trunk-calls - calling managers and distributors to note down orders. Hence, I was exposed to the excitement of targets, achievements, deficits and campaigns very early in my life. The idea of ‘work-life balance’ was not in vogue. One would look to their parents who worked long hours with pride making it something you wanted to emulate. Things have changed a lot but I would not trade the decision I made, to get into and stay in Pharma.
Do Retail Pharmacies Really Care about Patients?
The chemist shop—jammed between a chai stall and a...
Indian Oncology: An Unasked Question, the Challenges Ahead
A Brief History of Scale and an Unasked Question
In...
Are Quick Remedies Keeping Patients Away from Doctors?
The recent Mint Lounge analysis captured a striking consumer...
From Tragedy to Trust: What the CDCSO Can Learn from Dr. Frances Oldham Kelsey
Abstract
The U.S. Food and Drug Administration (FDA) evolved from...
Biocon’s Landmark Succession Planning: Claire Mazumdar is the Future Leader
In a landmark decision for India’s biotechnology sector, Kiran...
Do Retail Pharmacies Really Care about Patients?
The chemist shop—jammed between a chai stall and a...
Indian Oncology: An Unasked Question, the Challenges Ahead
A Brief History of Scale and an Unasked Question
In...
Are Quick Remedies Keeping Patients Away from Doctors?
The recent Mint Lounge analysis captured a striking consumer...
From Tragedy to Trust: What the CDCSO Can Learn from Dr. Frances Oldham Kelsey
Abstract
The U.S. Food and Drug Administration (FDA) evolved from...
Biocon’s Landmark Succession Planning: Claire Mazumdar is the Future Leader
In a landmark decision for India’s biotechnology sector, Kiran...
Do Retail Pharmacies Really Care about Patients?
The chemist shop—jammed between a chai stall and a...
Indian Oncology: An Unasked Question, the Challenges Ahead
A Brief History of Scale and an Unasked Question
In...
Are Quick Remedies Keeping Patients Away from Doctors?
The recent Mint Lounge analysis captured a striking consumer...
From Tragedy to Trust: What the CDCSO Can Learn from Dr. Frances Oldham Kelsey
Abstract
The U.S. Food and Drug Administration (FDA) evolved from...
Biocon’s Landmark Succession Planning: Claire Mazumdar is the Future Leader
In a landmark decision for India’s biotechnology sector, Kiran...
Do Retail Pharmacies Really Care about Patients?
The chemist shop—jammed between a chai stall and a...
Indian Oncology: An Unasked Question, the Challenges Ahead
A Brief History of Scale and an Unasked Question
In...
Are Quick Remedies Keeping Patients Away from Doctors?
The recent Mint Lounge analysis captured a striking consumer...
From Tragedy to Trust: What the CDCSO Can Learn from Dr. Frances Oldham Kelsey
Abstract
The U.S. Food and Drug Administration (FDA) evolved from...
Biocon’s Landmark Succession Planning: Claire Mazumdar is the Future Leader
In a landmark decision for India’s biotechnology sector, Kiran...
MedicinMan was launched in 2011 as India’s 1st Magazine dedicated to the cause of Field Force Excellence in Pharma and Devices.
Read More
Headlines
Do Retail Pharmacies Really Care about Patients?
The chemist shop—jammed between a chai stall and a...
Indian Oncology: An Unasked Question, the Challenges Ahead
A Brief History of Scale and an Unasked Question
In...
Are Quick Remedies Keeping Patients Away from Doctors?
The recent Mint Lounge analysis captured a striking consumer...
From Tragedy to Trust: What the CDCSO Can Learn from Dr. Frances Oldham Kelsey
Abstract
The U.S. Food and Drug Administration (FDA) evolved from...
Newsletter
Get important news delivered directly to your inbox and stay connected!