How the 2026 U.S. tariff action on branded drugs reshapes global pharma—and what it really means for Indian generics
Executive Summary
In April 2026, Donald Trump announced tariffs of up to 100% on imported patented (branded) pharmaceuticals, targeting companies that do not align with U.S. pricing expectations or expand domestic manufacturing. Crucially, generic medicines are exempt. For India—one of the largest suppliers of generics to the United States—this creates a paradox:
- Minimal direct exposure
- Potential competitive upside
- Significant long-term uncertainty
The policy is not designed to benefit India. Yet, by targeting high-cost innovator drugs, it may indirectly strengthen India’s position in the global generics market—if the industry responds strategically.
1. What the April 2026 Tariff Policy Actually Does


What the April 2026 Tariff Policy Actually Does
The April 2026 executive action is best understood as a targeted industrial and pricing intervention, not a blanket trade restriction.
Core Policy Objectives
- Drive down drug prices by linking U.S. prices to international benchmarks through “most-favoured-nation” (MFN) pricing mechanisms
- Incentivize domestic manufacturing by encouraging pharmaceutical companies to shift production of high-value drugs to the United States
Key Mechanism
- Up to 100% tariffs on non-compliant branded drug imports
- Lower tariffs for firms:
- Investing in U.S. manufacturing
- Entering pricing agreements
- Implementation timelines: ~120–180 days for compliance pathways
Explicit Exemptions
- Generic medicines
- Select low-cost essential drugs
- Certain specialized categories (e.g., orphan drugs)
This carve-out is not a concession—it is a system necessity.
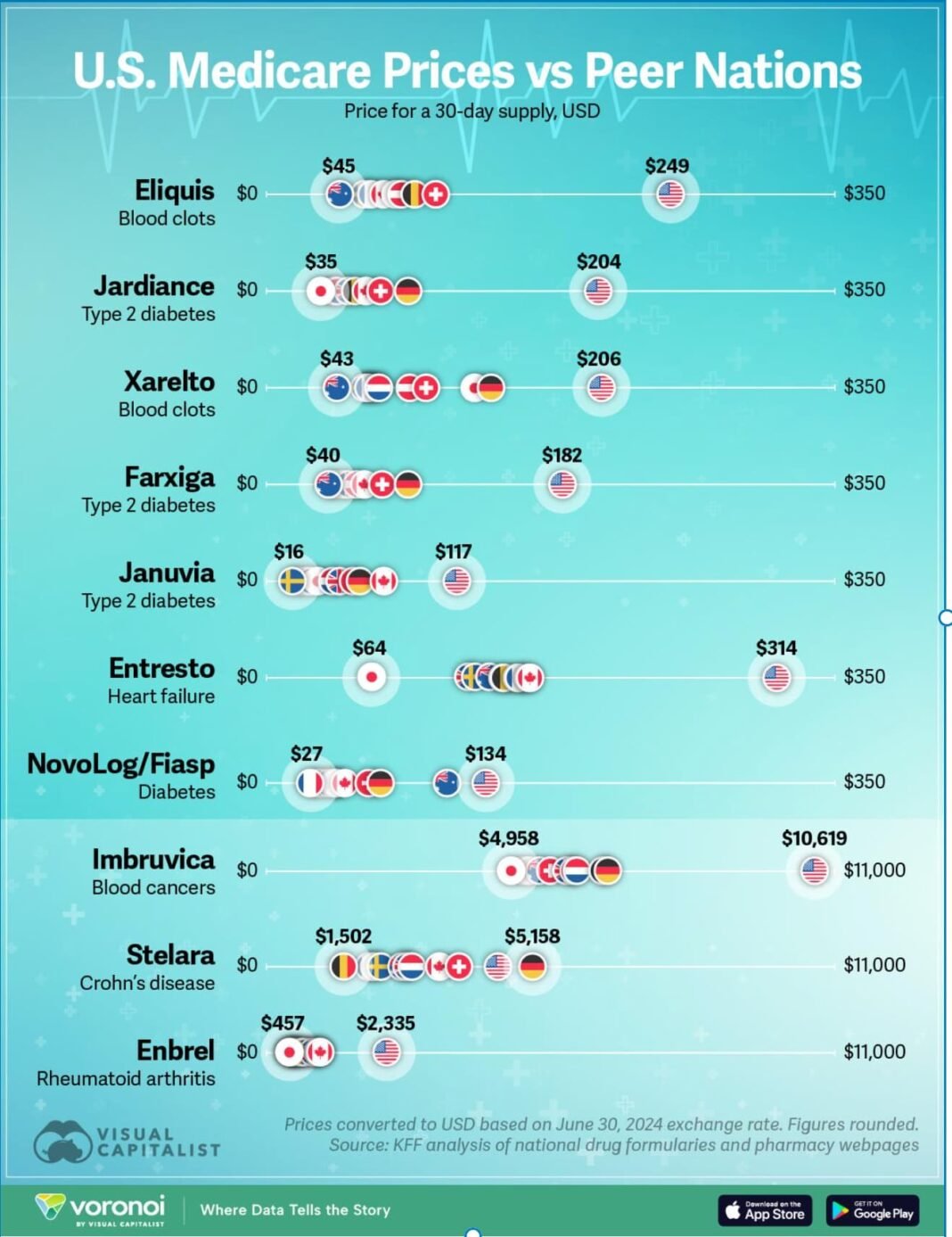
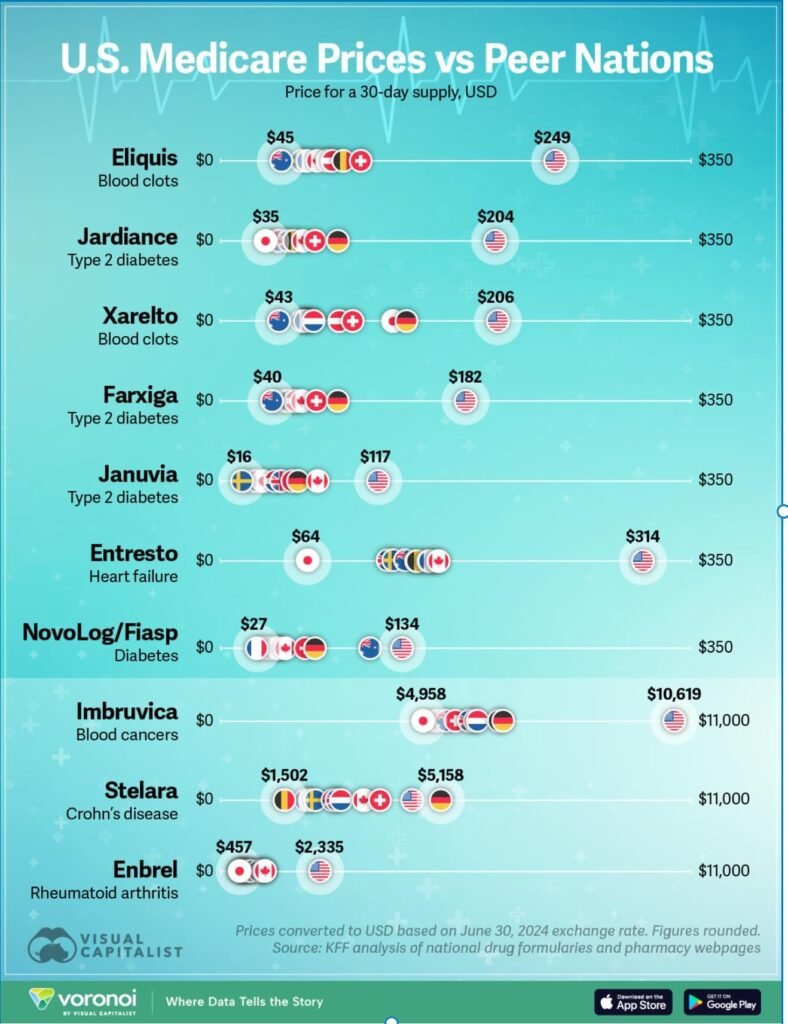
2. Why Generics Are Untouchable
The exclusion of generics reflects structural realities of the U.S. healthcare system.
a) Dominance in Volume
- Generics account for ~90% of prescriptions in the U.S.
- They represent a small fraction of total drug spending
b) Economic Constraint
Generic manufacturing operates on:
- Thin margins
- Globalized supply chains
Tariffs would:
- Raise prices for essential medicines
- Fail to meaningfully incentivize U.S. production
- Risk widespread shortages
c) System Dependence on India

India plays a central role:
- Supplies an estimated ~40–50% of U.S. generic medicines
- Enables hundreds of billions of dollars in annual healthcare savings
- Anchors supply for chronic therapies (cardiac, diabetes, anti-infectives)
This creates a “too critical to disrupt” dynamic.
3. India’s Position: Insulated—but Not Immune
a) Export Profile Advantage
India’s U.S. pharma exports are overwhelmingly generic, not patented.
Implication:
→ The bulk of exports falls outside tariff scope
b) Immediate Impact
- Negligible direct tariff exposure
- Limited impact on:
- Specialty portfolios
- Niche branded products
c) Structural Leverage
India’s role is not just as a supplier—but as a system stabilizer in U.S. healthcare economics.
4. Who Actually Gets Hit
The policy primarily targets:
| Segment | Impact |
|---|---|
| Multinational innovator pharma | High |
| Offshore patented drug manufacturing | High |
| Generic manufacturers | Minimal (exempt) |
| API supply chains | Under review |
This creates a relative competitive shift, not an absolute advantage.
5. The India–U.S. Trade Context: Reality vs Narrative
What is supported:
- Ongoing India–U.S. trade engagement
- Signals of continued pharmaceutical exemptions
- Likely negotiated outcomes under U.S. national security reviews (Section 232)
What is NOT fully substantiated:
- A formal, binding “0% tariff deal” exclusively protecting Indian generics
- Guaranteed long-term preferential access
👉 Conclusion:
India’s position is policy-enabled, not treaty-locked.
6. The Real Opportunity for Indian Pharma
1) Short-Term: Demand Substitution
Higher costs for branded drugs may:
- Accelerate generic substitution
- Increase volume demand for Indian exporters
2) Supply Chain Realignment
Geopolitical pressures—especially U.S.–China tensions—create tailwinds:
- Diversification away from China
- Increased trust in India as a reliable supplier
3) Margin Stability (Not Windfall)
Unlike the earlier draft’s assumption of large margin expansion:
- Gains are likely to be incremental
- Driven by volume, not pricing power
4) Strategic Window for Value Migration
The real upside lies beyond generics:
- Complex generics
- Biosimilars
- Specialty and differentiated products
This aligns with India’s long-term ambition to shift from: “Pharmacy of the world” → “Innovation-led pharma power”
7. The Critical Risk Layer
1) Section 232: The Unresolved Variable
The U.S. continues to evaluate pharmaceutical imports under national security provisions. Potential future focus:
- APIs
- Critical therapies
- Supply chain dependencies
👉 This is the single biggest policy risk.
2) API Dependence on China
India still relies significantly on China for:
- Bulk drugs
- Key starting materials
Despite PLI schemes, this remains a strategic vulnerability.
3) Policy Volatility in the U.S.
Recent developments show:
- Frequent legal challenges to tariff authority
- Use of alternative trade instruments
👉 Expect continued unpredictability
4) Quality and Regulatory Scrutiny
- India has the largest number of USFDA-approved plants outside the U.S.
- Yet:
- Warning letters
- Import alerts
remain persistent risks.
Quality is not a differentiator—it is a license to operate.
8. Strategic Interpretation
This policy should not be misread as protectionism against global pharma. It is:
A targeted intervention against high-priced innovator drugs, driven by domestic political and economic priorities.
For India:
- The benefit is indirect
- The opportunity is time-bound
- The outcome depends on execution
Conclusion: A Window, Not a Windfall
The April 2026 tariff action does not fundamentally reorder global pharma trade—but it does create temporary asymmetry. India’s generics sector is:
- Protected by necessity
- Positioned for incremental gains
- Exposed to future policy shifts
The real question is not whether India benefits. It is whether Indian pharma can use this window to:
- Reduce API dependence
- Strengthen quality systems
- Move up the value chain
Because tariff advantages fade. Capability advantages endure.

Sources and References
- Reuters
“Drugmakers face 100% tariff unless they cut prices or produce drugs in US”
April 2, 2026
https://www.reuters.com/sustainability/boards-policy-regulation/drugmakers-face-100-tariff-unless-they-cut-prices-or-produce-drugs-us-2026-04-02/ - Associated Press
Coverage of U.S. pharmaceutical tariff announcement and compliance timelines
April 2026
https://apnews.com/ - The Wall Street Journal
“Trump administration unveils up to 100% tariff on branded drugs”
April 2026
https://www.wsj.com/ - The Guardian
“Trump announces tariffs on drug imports to pressure pharma companies”
April 2, 2026
https://www.theguardian.com/us-news/2026/apr/02/trump-100-tariff-us-pharmaceutical-drug-makers - Barron’s
Analysis of tariff bands and global pharma implications
April 2026
https://www.barrons.com/
2. U.S. Policy and Legal Framework
- U.S. Department of Commerce
Section 232 Investigations Overview
https://www.commerce.gov/ - U.S. Trade Representative
Trade policy and tariff frameworks
https://ustr.gov/ - United States Congress
Trade Expansion Act of 1962 (Section 232)
https://www.congress.gov/ - Supreme Court of the United States
Recent rulings affecting executive tariff authority (2026 context)
https://www.supremecourt.gov/
3. U.S. Pharmaceutical Market Structure
- IQVIA
The Use of Medicines in the U.S. – Generics share and cost savings
https://www.iqvia.com/ - U.S. Food and Drug Administration
Generic Drug Facts – Utilization and approvals
https://www.fda.gov/drugs/generic-drugs/generic-drug-facts - Association for Accessible Medicines
U.S. Generic Drug & Biosimilar Savings Report
https://accessiblemeds.org/resources/reports
4. India’s Role in Global Generics
- Indian Pharmaceutical Alliance
Statements on U.S. dependence on Indian generics
https://www.ipa-india.org/ - Ministry of Commerce and Industry India
Pharmaceutical export data and trade positioning
https://commerce.gov.in/ - Pharmexcil
India pharma export statistics
https://pharmexcil.com/
5. India–U.S. Trade Context
- The Times of India
“Interim pact brings certainty and clarity for pharma sector”
February 2026
https://timesofindia.indiatimes.com/ - Financial Express
Coverage of India–U.S. trade discussions impacting pharma
https://www.financialexpress.com/ - CNBC TV18
Policy commentary and ministerial statements
https://www.cnbctv18.com/
6. Industry and Policy Analysis
- Pharmaphorum
Impact of U.S. tariffs on global pharma supply chains
https://pharmaphorum.com/ - Cyril Amarchand Mangaldas
Legal analysis of pharma tariffs and implications for India
https://disputeresolution.cyrilamarchandblogs.com/ - Brookings Institution
Drug pricing and global pharmaceutical policy analysis
https://www.brookings.edu/
7. Supply Chain and API Dependence
- Department of Pharmaceuticals India
PLI Scheme for Bulk Drugs and API localization
https://pharmaceuticals.gov.in/ - NITI Aayog
Reports on API dependence and domestic manufacturing strategy
https://www.niti.gov.in/ - World Health Organization
Global pharmaceutical supply chain insights
https://www.who.int/
8. Additional Contextual References
- World Trade Organization
Tariff principles and trade frameworks
https://www.wto.org/ - OECD
Pharmaceutical pricing and market access studies
https://www.oecd.org/
Notes on Source Use
- All quantitative claims in the article are derived from IQVIA, FDA, or industry bodies
- Policy interpretations are based on Reuters, WSJ, and government frameworks
- Trade context reflects reported developments, not legally binding treaty claims

All Images are AI Generated for Illustration Only. E&OE