“Lifestyle advice often asks patients to fight powerful evolutionary biology indefinitely.”
“For the first time, pharmacology can significantly alter the biological signals that regulate appetite.”
“These drugs do not merely suppress hunger—they modify the metabolic pathways that generate it.”
“The future of treating lifestyle disease may lie in aligning biology with behaviour.”
Are Drugs That Rewire the Brain the Future of Treating Lifestyle Diseases?
For decades the prescription for lifestyle diseases has been simple and familiar: eat less, exercise more, sleep better, reduce stress.
The advice is correct. Yet the global rise of conditions such as Obesity, Type 2 Diabetes, and Hypertension suggests that correct advice alone is not enough.
Patients generally know what healthier living requires. The difficulty lies in sustaining behavioural change over years or decades. In practice, lifestyle change resembles learning a new language: many start enthusiastically, but relatively few persist long enough to achieve lasting fluency.
The Counter-Argument: Are We Medicalising Lifestyle?
The rise of metabolic drugs has also sparked debate.
Some physicians worry that widespread use could medicalise lifestyle disorders, shifting attention away from diet, physical activity, and broader social determinants of health.
Cost is another issue. Many of these therapies remain expensive, raising concerns about long-term affordability and healthcare equity.
Safety monitoring also continues. While clinical trials have shown encouraging results, researchers are still evaluating potential long-term effects involving the pancreas, gastrointestinal system, and muscle mass.
These concerns underline the importance of balanced adoption.

Now a new class of medicines is challenging long-standing assumptions about how lifestyle diseases should be treated.
Drugs such as Semaglutide and Tirzepatide, developed by Novo Nordisk and Eli Lilly, do something earlier therapies could not: they directly influence the biological systems that regulate appetite and metabolism.
This raises an increasingly important question for clinicians:
If biology drives appetite so strongly, could drugs that alter these signals become the primary treatment for lifestyle diseases?
The Deeper Shift
Perhaps the most important change is conceptual.
Obesity and related conditions are gradually being understood not simply as behavioural problems but as complex neuro-metabolic disorders involving hormones, brain pathways, and energy regulation.
If that perspective continues to gain acceptance, pharmacological therapy may become a routine component of managing lifestyle diseases.
Drugs may not eliminate the need for discipline.
But they may finally reduce the biological resistance that has made discipline so difficult.

The Biological Limits of Discipline
For many years obesity was framed largely as a matter of personal responsibility. Patients were urged to reduce calorie intake and increase physical activity.
Modern physiology has revealed why that advice, though well intentioned, often fails.
When calorie intake falls, the body activates several defence mechanisms:
- hunger hormones increase
- metabolic rate slows
- neural reward pathways intensify the drive to eat
These responses evolved to protect humans against starvation. Unfortunately, in modern environments rich in calories, they make sustained weight loss extremely difficult.
Lifestyle advice therefore asks patients to fight powerful evolutionary biology indefinitely.

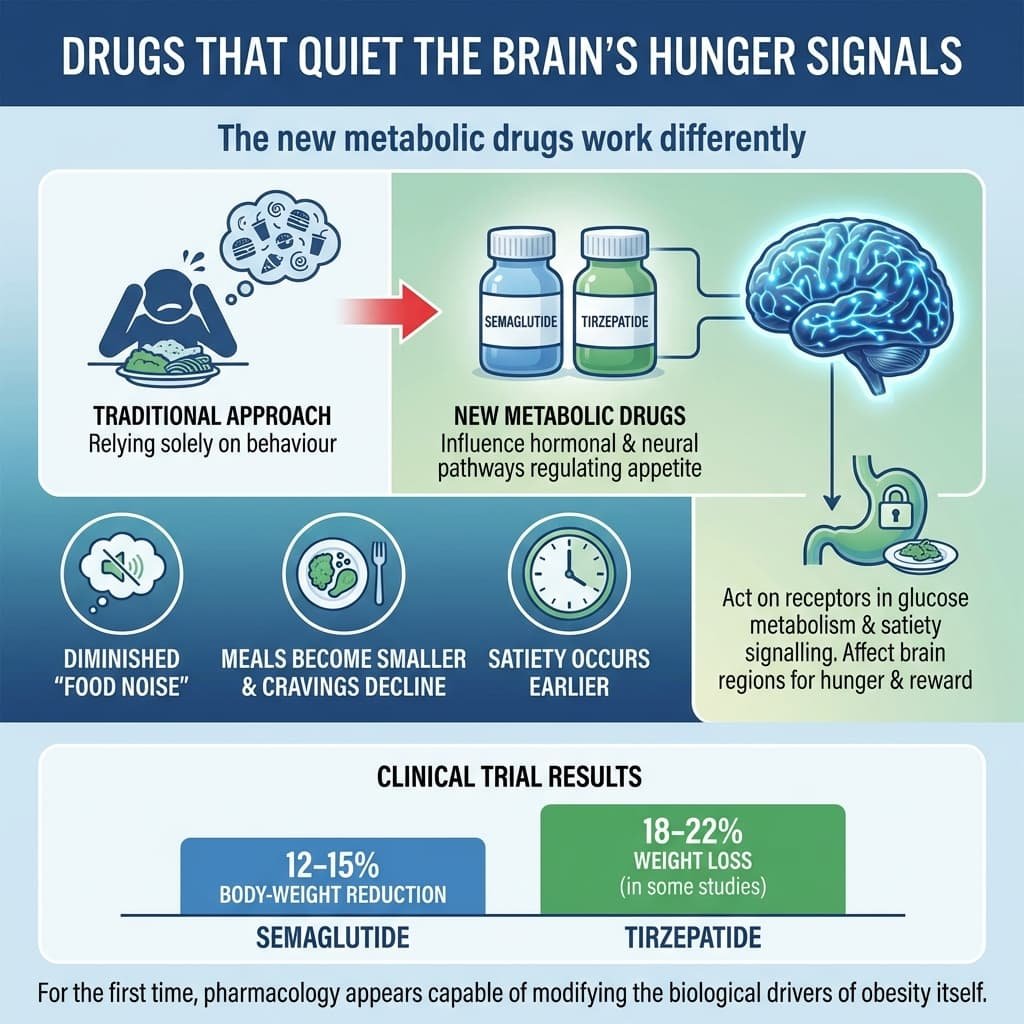
Drugs That Quiet the Brain’s Hunger Signals
The new metabolic drugs work differently.
Instead of relying solely on behaviour, they influence the hormonal and neural pathways that regulate appetite.
Both semaglutide and tirzepatide act on receptors involved in glucose metabolism and satiety signalling. Importantly, they also affect brain regions responsible for hunger and reward.
Many patients describe a striking change: the constant mental “noise” about food diminishes. Meals become smaller, cravings decline, and satiety occurs earlier.
Clinical trials have shown results once associated mainly with Bariatric surgery.
- Semaglutide can produce 12–15% body-weight reduction
- Tirzepatide has demonstrated 18–22% weight loss in some studies
For the first time, pharmacology appears capable of modifying the biological drivers of obesity itself.
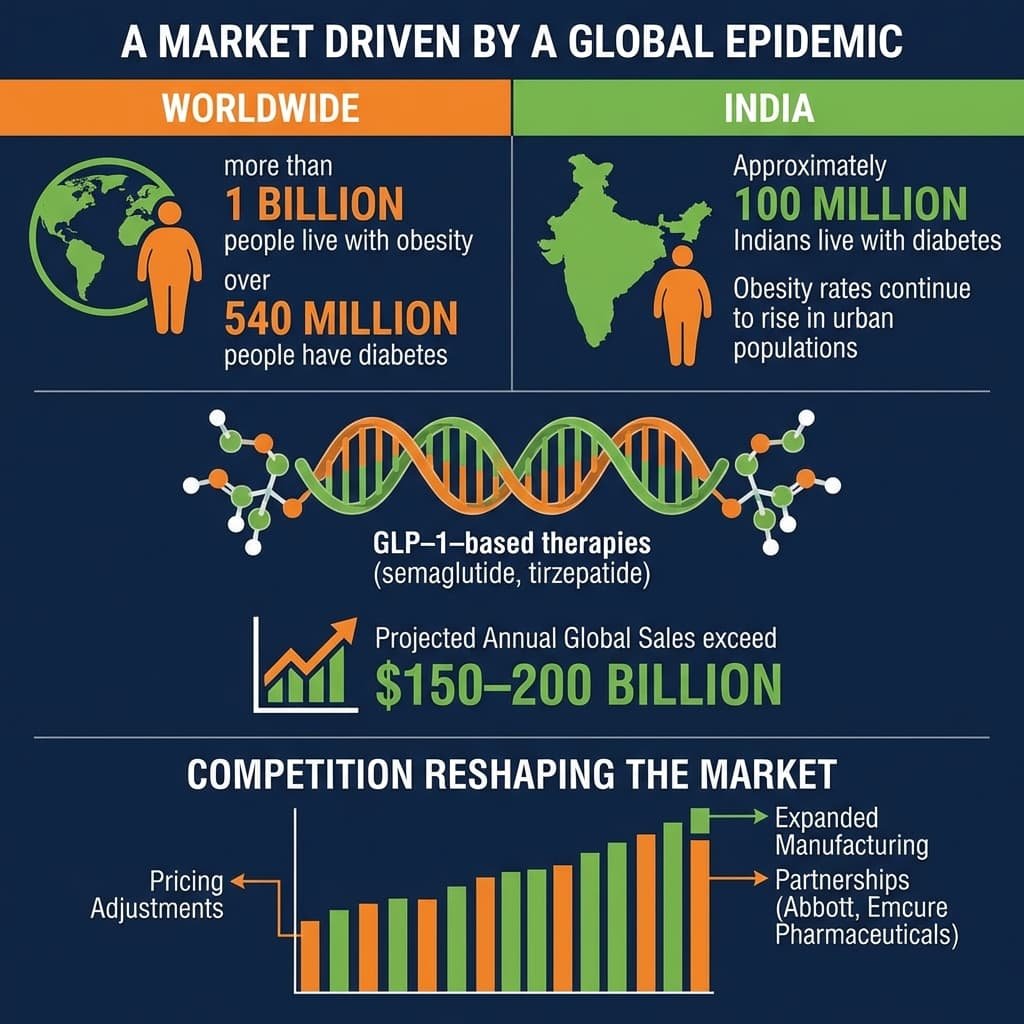
A Market Driven by a Global Epidemic
The scale of metabolic disease makes the implications enormous.Worldwide:
- more than 1 billion people live with obesity
- over 540 million people have diabetes
India reflects the same pattern. Approximately 100 million Indians live with diabetes, while obesity rates continue to rise in urban populations.
Because drugs like semaglutide and tirzepatide influence both glucose metabolism and body weight, they sit at the centre of multiple chronic diseases.
Industry analysts now expect GLP-1–based therapies and related drugs to become one of the largest pharmaceutical markets in history, with projections suggesting annual global sales could exceed $150–200 billion within the next decade.
Competition is already reshaping the market. Pricing adjustments, expanded manufacturing, and partnerships with companies such as Abbott and Emcure Pharmaceuticals are helping extend access beyond specialised endocrinology clinics.
Key Numbers — The Metabolic Disease Landscape
Global obesity: ~1 billion people
Global diabetes: ~540 million people
Weight loss in trials
- Semaglutide: 12–15% body weight
- Tirzepatide: 18–22% body weight
Projected global GLP-1 market:
$150–200 billion annually within a decade
Estimated people with diabetes in India: ~100 million
A New Model of Treatment
The future may not involve replacing lifestyle medicine but reordering it.
Instead of waiting years for lifestyle interventions to succeed before prescribing drugs, clinicians may increasingly adopt a combined approach: early pharmacological therapy alongside sustained behavioural change.
In this model, medicines stabilise the biological drivers of appetite while lifestyle choices determine long-term health outcomes.


All Images are AI Generated for Illustration Only. E&OE
Selected Sources
• World Health Organization — Global obesity and metabolic disease reports
• International Diabetes Federation — Global diabetes prevalence data
• American Diabetes Association — Standards of care in diabetes and obesity
• The New England Journal of Medicine — Clinical trials of semaglutide and tirzepatide
• The Lancet — Global obesity research
• Nature Medicine — Reviews on appetite regulation and neuro-metabolic signalling