The Paradox of Protection
In the time it takes to read this sentence, roughly 500 children worldwide receive a life-saving vaccine. Many carry the branding of Western giants, yet the doses that reach the Global South are overwhelmingly produced by manufacturers in India and China. This is not a flaw in the system—it is the system: a carefully balanced architecture of high-margin innovation in the West and high-volume affordability in the South.
The global vaccine market hit approximately $82 billion in 2025 (Cognitive Market Research, December 2025 update). While commercial estimates capture premium pricing in high-income markets, the WHO’s 2025 Global Vaccine Market Report (data for 2024) records a “financial value” of $66.5 billion across 7.2 billion doses—highlighting the stark reality of tiered pricing that makes vaccines accessible where they are needed most.
This investigation draws on FY2024-25 company filings, UNICEF/Gavi procurement data, WHO prequalification records, and independent market analyses to profile the true architects: the United States, India, China, the United Kingdom, France, and emerging niche players.
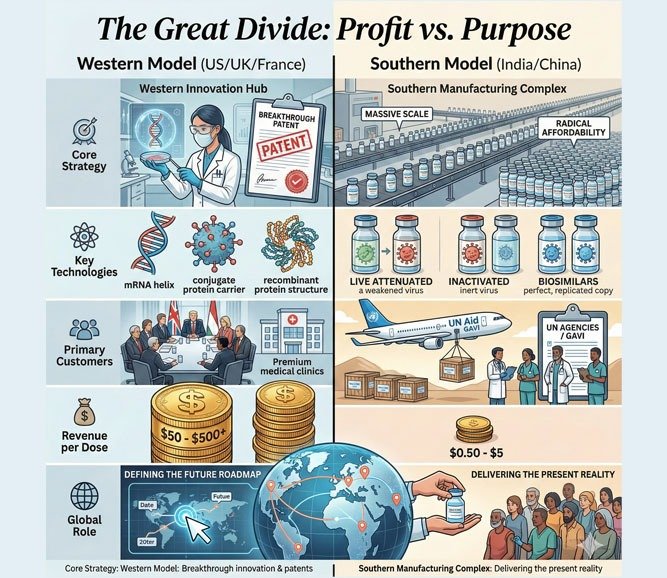
The Great Divide: Profit vs. Purpose
| Metric | Western Model (US/UK/France) | Southern Model (India/China) |
| Core Strategy | Breakthrough innovation & patents | Massive scale & radical affordability |
| Key Technologies | mRNA, conjugate, recombinant | Live attenuated, inactivated, biosimilars |
| Primary Customers | G7 governments, private markets, insurers | UN agencies, Gavi, LMICs, bilateral deals |
| Revenue per Dose | $50–$500+ | $0.50–$5 |
| Global Role | Defining the future roadmap | Delivering the present reality |
This engineered divide—rooted in intellectual property frameworks, regulatory pathways, and procurement policies—means dominance flips depending on the metric you choose: dollars favor the West, doses favor the South.
Revenue vs. Volume Snapshot (FY2024-25 estimates)
| Nation/Group | Approx. Revenue | Approx. Volume (doses) | Primary Strength |
| United States | $23 billion | 600 million | High-value innovation |
| United Kingdom | $10–11 billion | 250 million | Premium adult & respiratory |
| France | $8 billion | 350 million | Routine EPI backbone |
| India (firms) | $4–5 billion | 2.5–3 billion | Volume king for LMICs |
| China (firms) | $3 billion | 1.5–2 billion | Domestic security + diplomacy |
| Others (incl. SK, DK, JP) | ~$2–3 billion | ~300 million | Niche specialty & resilience |
United States: The Innovation Engine
Revenue (FY2024-25): ~$23 billion | Volume: ~600 million doses
Pfizer, Merck, and Moderna continue to dominate high-value segments. Merck’s Gardasil franchise remains the cornerstone of global cervical-cancer elimination efforts. Pfizer-BioNTech and Moderna have pivoted successfully to RSV, flu, and combination respiratory vaccines using mRNA platforms. These products command premium pricing in wealthy markets but face access barriers in low- and middle-income countries—placing greater reliance on Gavi donors.
India: The Pharmacy of the World
Revenue (FY2024-25): $4–5 billion | Volume: 2.5–3 billion doses
Serum Institute of India alone sells approximately 1.5 billion doses annually (capacity now exceeding 4 billion), supplying pentavalent, DPT, measles-rubella, and pneumococcal vaccines to UNICEF tenders at under $2 per dose. Bharat Biotech, Biological E, and others round out the ecosystem. India’s polio eradication journey and its pivotal COVAX role demonstrated both its indispensable scale and the supply-chain risks of over-reliance on a single geography—prompting healthy diversification into Africa and beyond.
China: The Dual-Power Player
Revenue (FY2024-25): ~$3 billion | Volume: 1.5–2 billion doses
Sinovac, CanSino, and CNBG have achieved near-total domestic coverage for key diseases such as hepatitis B while expanding bilateral supply across Asia and Africa. China now accounts for roughly half the financial value of the global HPV market in the private sector and is rapidly advancing inhaled and mRNA platforms—signaling a serious challenge to Western technological leadership.
United Kingdom: Research Powerhouse with Global Reach
Revenue (FY2024-25): $10–11 billion | Volume: ~250 million doses
GSK’s Shingrix (shingles) and respiratory portfolio, combined with AstraZeneca’s legacy equity model, keep the UK at the forefront. Oxford-AstraZeneca’s at-cost COVID-19 supply during the pandemic set a precedent that still influences global expectations around equitable access.
France: The Quiet Essential
Revenue (FY2024-25): ~$8 billion | Volume: ~350 million doses
Sanofi remains the indispensable supplier of DTP, polio, and routine EPI vaccines that keep national immunization programs running smoothly worldwide. Valneva’s chikungunya vaccine (approved 2023) exemplifies Europe’s ability to address emerging climate-driven threats.
Niche Protectors: Denmark, South Korea, and Japan
Denmark: ~$0.5 billion | ~25 million doses
Bavarian Nordic’s smallpox/mpox stockpiles serve as a critical global security net.
South Korea has surged with SK Bioscience and others, capturing a growing share of the Asian market (~$2.2 billion regional in 2025 per Cognitive). Japan’s Daiichi Sankyo and partners add further resilience in high-tech segments.
Together these players prove that specialized excellence and regional self-reliance matter as much as sheer scale.
Synthesis: A Fragile but Remarkable Architecture
The United States and United Kingdom push the technological frontier. India and China deliver the doses that actually reach billions. France and European partners keep routine immunization alive. Niche players safeguard against outliers.
Yet vulnerabilities remain: over-concentration of certain APIs, Gavi’s funding dependence for many Indian suppliers, and the fragility exposed by COVID-19 supply shocks. Promising counter-trends include Africa’s Afrigen mRNA technology transfer (aiming for 60% regional self-sufficiency by 2030) and innovative hybrids such as Biological E’s Novavax partnership.
The Architects’ Responsibility
These nations do not merely produce vaccines—they define global health security. Future victories against malaria, TB, and the next inevitable pandemic will require deliberate hybrid models: Western innovation paired with Southern scale, transparent technology transfer, and pricing that reflects both R&D risk and equity imperatives.
The architecture is already in place. The question is whether we will strengthen its foundations—or allow short-term pressures to crack them.
Appendix: Credible Sources (Direct Access)
Market Reports & Official Data
- Cognitive Market Research – Vaccine Market Report 2025 (updated Dec 2025) – https://www.cognitivemarketresearch.com/vaccine-market-report
- WHO Global Vaccine Market Report 2025 (data for 2024) – https://cdn.who.int/media/docs/default-source/immunization/mi4a/2025-who-global-vaccine-market-report.pdf
- UNICEF Vaccine Product Prices & Procurement Data – https://www.unicef.org/supply/vaccines-pricing-data
- Gavi, the Vaccine Alliance – Annual Progress Reports & Factsheets
Company & Financial Disclosures 5. Pfizer, Merck, Moderna – SEC EDGAR 10-K filings (2024-2025) 6. GSK Full-Year 2025 Results – https://www.gsk.com 7. Sanofi Full-Year 2025 Results – https://www.sanofi.com 8. Serum Institute of India – Corporate disclosures & press releases (2024-2025)
Specialized Analyses 9. Fierce Pharma – 2024-2025 Vaccine Sales Rankings 10. Evaluate Pharma / Statista – Global Vaccine Company Revenue Rankings 11. Grand View Research – India & China Vaccine Market Outlooks (2025) 12. id-ea.org – “The global vaccine market 2024-2025” (company dose & revenue estimates)
Additional Context 13. WHO Prequalification of Vaccines – Public Database 14. OANDA Historical Currency Conversions (for USD standardization)
All figures represent best-available triangulated estimates as of Q1 2026. Private-company volume and revenue data are approximations drawn from capacity disclosures, tender wins, and audited filings where available.
© 2026 MedicinMan Publications. For updates or data queries: editorial@medicinman.net