On February 13, 2026, the Pharmacovigilance Risk Assessment Committee (PRAC) of the European Medicines Agency recommended the withdrawal of all levamisole-containing medicines from the European Union market.
The trigger was not a minor safety update. It was a rare but potentially devastating neurological condition — leukoencephalopathy, a disorder affecting the brain’s white matter. After reviewing accumulated pharmacovigilance data, PRAC concluded that the risk could not be adequately mitigated through warnings, dose adjustments, or monitoring. The benefit–risk balance, in their assessment, had turned negative.
For a molecule introduced in Europe in 1966 as an anthelmintic, this marks a decisive regulatory endpoint.
For India, where levamisole remains available and in active use, the decision presents something more consequential: a test of regulatory maturity.
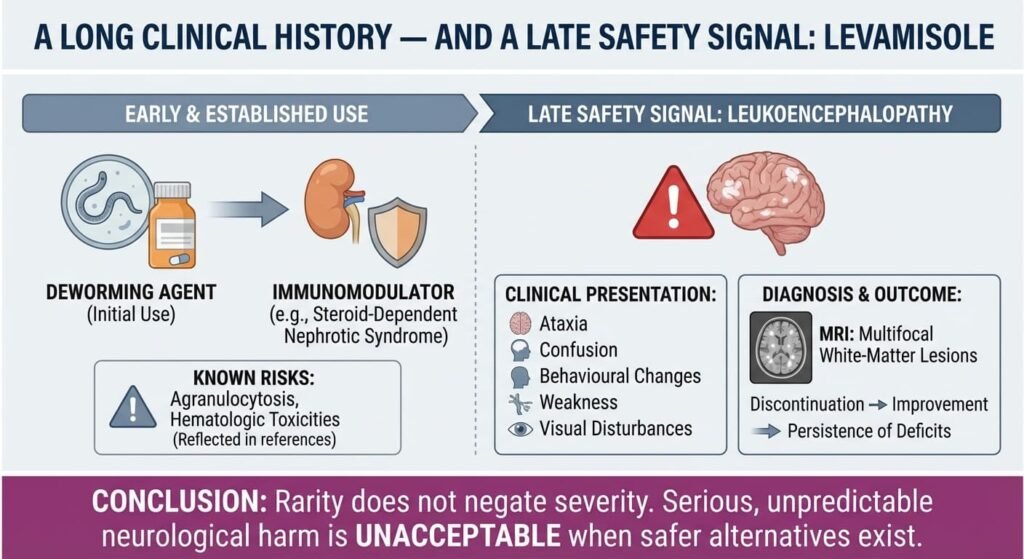
A Long Clinical History — and a Late Safety Signal
Levamisole began as a deworming agent and later found use as an immunomodulator, including in steroid-dependent nephrotic syndrome. Over decades, its known risks included agranulocytosis and other hematologic toxicities — adverse effects already reflected in Indian prescribing references. What has changed is the recognition of levamisole-associated leukoencephalopathy.
Clinically, patients may present with ataxia, confusion, behavioural changes, weakness, or visual disturbances. MRI typically reveals multifocal white-matter lesions. In some cases, symptoms improve after discontinuation; in others, neurological deficits may persist.
The rarity of the condition makes it difficult to detect in routine post-marketing surveillance — but rarity does not negate severity. The European review concluded that even infrequent, unpredictable, serious neurological harm was unacceptable when safer therapeutic alternatives exist.
The Indian Context: What Is Different — and What Is Not
Levamisole continues to be listed in Indian drug compendia such as CIMS and remains available as a low-cost generic through retail channels.
It is important to clarify: levamisole is not part of India’s National Deworming Day programme, which relies on albendazole. However, it continues to be prescribed in individual clinical settings, including for helminth infections and certain immunomodulatory indications.
At present, there is limited publicly available Indian pharmacovigilance data specifically examining levamisole-associated leukoencephalopathy. The absence of publicly reported clusters does not confirm absence of risk — particularly for delayed, rare neurological events.
This is where the issue becomes systemic rather than drug-specific.
Pharmacovigilance: A Structural Challenge
India’s Adverse Drug Reaction monitoring is coordinated through the National Coordination Centre – Pharmacovigilance Programme of India (NCC-PvPI) at the Indian Pharmacopoeia Commission, Ghaziabad. Reporting volumes have improved in recent years, and awareness among healthcare professionals is increasing.
However, rare, delayed, or low-frequency neurological syndromes present a unique challenge:
• They may occur weeks or months after exposure.
• They may be misattributed to infections, autoimmune disorders, or demyelinating disease.
• They require imaging correlation and specialist evaluation.
• They depend heavily on clinician suspicion to trigger reporting.
Passive spontaneous reporting systems are structurally weaker at detecting precisely this type of signal.
The levamisole case therefore raises a larger question: Can India independently detect and evaluate rare but severe safety signals without relying on decisions made by higher-resourced regulators?

Regulatory Alignment or Regulatory Gap?
India’s drug regulator, the Central Drugs Standard Control Organization (CDSCO), has introduced important reforms in recent years, particularly through amendments to the New Drugs and Clinical Trials Rules to streamline approvals.
But accelerated approval pathways must be matched by equally robust post-marketing review.
When a major regulator such as the European Medicines Agency determines that a long-standing drug no longer meets acceptable safety thresholds, it creates a decision point for other jurisdictions:
• Conduct an independent reassessment.
• Request local data.
• Update labeling.
• Restrict use.
• Or maintain status quo.
Maintaining status quo without formal review risks creating a two-tier safety standard — one for high-surveillance markets, another for lower-signal environments.
That perception alone can affect professional confidence.

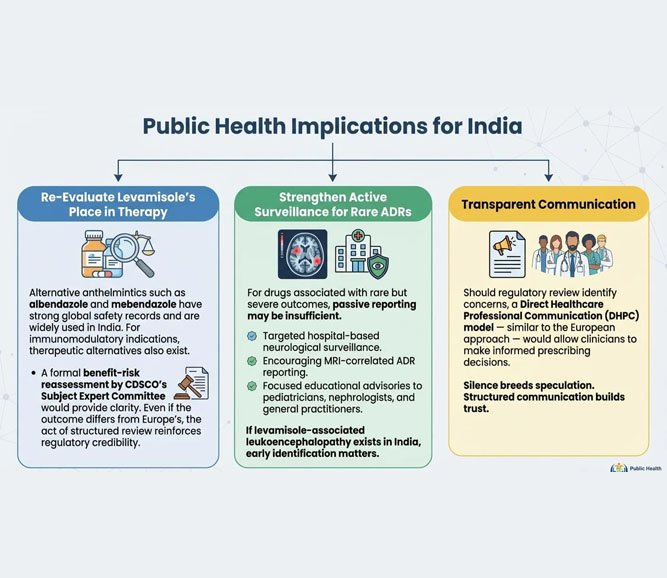
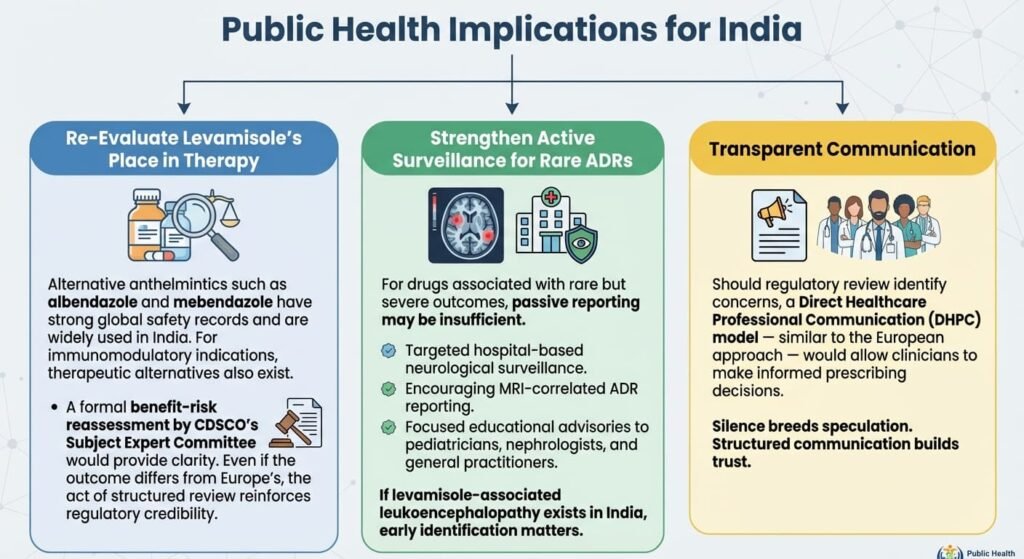
Public Health Implications for India
- Re-Evaluate Levamisole’s Place in Therapy
Alternative anthelmintics such as albendazole and mebendazole have strong global safety records and are widely used in India. For immunomodulatory indications, therapeutic alternatives also exist.
A formal benefit–risk reassessment by CDSCO’s Subject Expert Committee would provide clarity. Even if the outcome differs from Europe’s, the act of structured review reinforces regulatory credibility.
- Strengthen Active Surveillance for Rare ADRs
For drugs associated with rare but severe outcomes, passive reporting may be insufficient. Consider:
• Targeted hospital-based neurological surveillance.
• Encouraging MRI-correlated ADR reporting.
• Focused educational advisories to pediatricians, nephrologists, and general practitioners.
If levamisole-associated leukoencephalopathy exists in India, early identification matters.
- Transparent Communication
Should regulatory review identify concerns, a Direct Healthcare Professional Communication (DHPC) model — similar to the European approach — would allow clinicians to make informed prescribing decisions.
Silence breeds speculation. Structured communication builds trust.
The Larger Question: The levamisole episode is not fundamentally about one drug.
It is about whether India’s pharmacovigilance architecture can:
• Detect rare neurological harms,
• Analyse domestic data independently,
• Act proportionately and transparently,
• And communicate decisions clearly to clinicians and the public.
If a reassessment concludes that levamisole’s benefits remain acceptable in defined indications, that conclusion should be publicly justified with data. If not, withdrawal should be proactive — not reactive. Regulatory maturity is measured not by how quickly drugs are approved, but by how decisively emerging risks are addressed. The real question is not whether Europe withdrew levamisole. The real question is whether India’s regulatory system can independently determine — and defend — its own answer.
Sources
Regulatory Decision
1. European Medicines Agency
“EMA recommends withdrawal of levamisole medicines due to risk of leukoencephalopathy.”
February 13, 2026.
Official press release and PRAC recommendation.
2. PRAC Meeting Highlights — February 2026
Official committee summary documenting benefit–risk assessment.
Indian Clinical Context
3. CIMS India — Levamisole Monograph
Current Indian prescribing information, indications, dosing, and listed adverse effects.
4. Apollo Pharmacy Drug Information Portal — Levamisole
Patient-facing safety and warning details reflecting Indian market labeling.
Pharmacovigilance & Regulatory Framework
5. National Coordination Centre – Pharmacovigilance Programme of India (NCC-PvPI), Indian Pharmacopoeia Commission, Ghaziabad
Suspected ADR Reporting Portal and Programme Overview.
6. Review Article: Pharmacovigilance in India — Challenges and Future Directions
Peer-reviewed open-access analysis of ADR reporting infrastructure and limitations.
7. Central Drugs Standard Control Organization
New Drugs and Clinical Trials Rules (latest amendments) — regulatory framework for post-approval oversight.

Editorial Note: Time for a Formal CDSCO Review on Levamisole
The recent recommendation by the European Medicines Agency to withdraw levamisole from the European market is not merely a regional regulatory development. It is a signal.
When a medicine with nearly six decades of clinical use is reassessed and found to carry an unacceptable neurological risk, every jurisdiction where that medicine remains in use has a responsibility to examine its own data, context, and benefit–risk balance.
Levamisole continues to be available in India. At present, there is no publicly announced review by the Central Drugs Standard Control Organization regarding its safety profile in light of the European decision.
MedicinMan believes that a formal, transparent reassessment by CDSCO’s Subject Expert Committee is both prudent and necessary. Such a review need not presuppose withdrawal. It should, however, evaluate:
• Available Indian pharmacovigilance data
• Global safety findings
• Therapeutic alternatives
• The drug’s current place in therapy
Regulatory confidence is strengthened not by silence, but by visible, evidence-based evaluation.
India has built significant pharmacovigilance capacity over the past decade. This is an opportunity to demonstrate that the system can independently assess emerging risks and communicate decisions clearly to healthcare professionals.
Patient safety must remain the anchor.
MedicinMan will continue to follow developments closely and encourages clinicians and stakeholders to engage constructively in this discussion.

All Images are AI Generated for Illustration Only. E&OE