The era of Chinese biotech as a mere imitator is history; the world must now reckon with China as a true leader in drug innovation
China’s biotechnology sector has burst onto the global stage, transforming from a maker of generic drugs into a powerful force challenging Western dominance in pharmaceutical innovation. Here’s how this “Made in China Biotech” revolution is rewriting the rules.
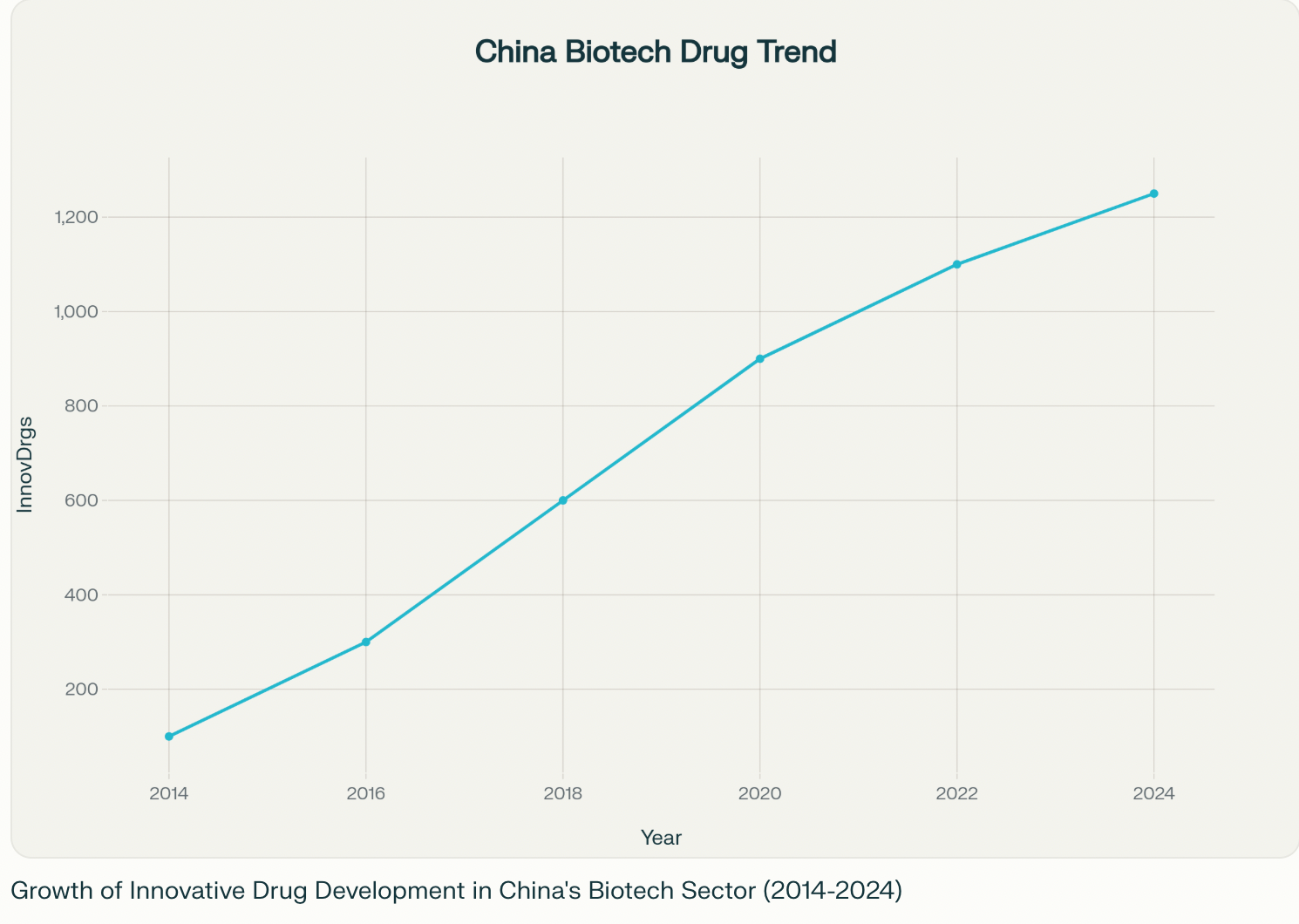
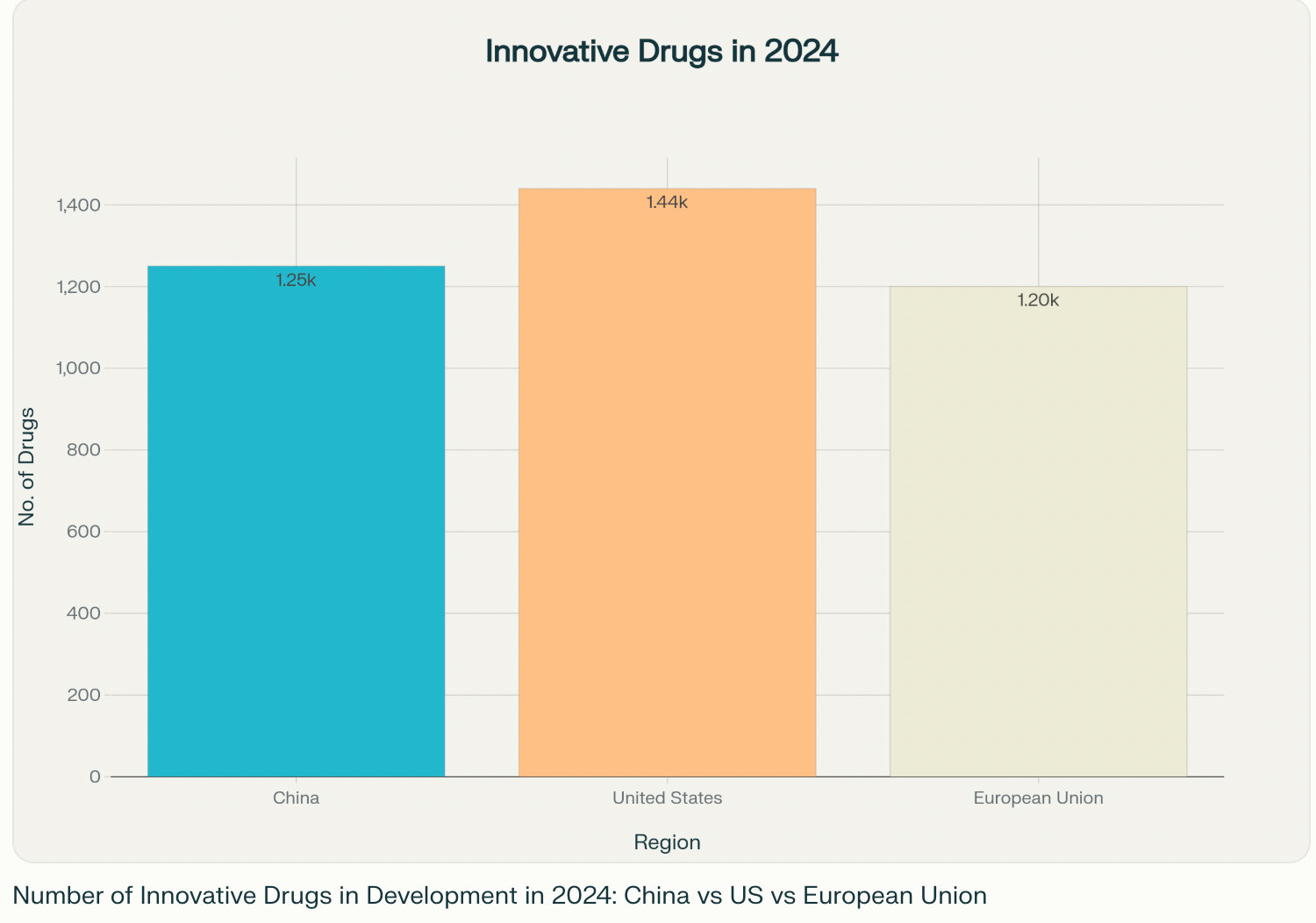
From Imitator to Innovator
- Breakneck Progress: In 2024, over 1,250 innovative drugs—from cancer treatments to cutting-edge therapies—entered development in China, surpassing the European Union and coming close to the U.S. total of about 1,440. Just a decade ago, these numbers seemed impossible for a country once known mostly for producing generic medicines123.
- Quality Leap: Chinese biopharma isn’t just about scale, but surging quality. Homegrown companies are now winning fast-track designations from top regulators like the U.S. FDA and EMA for breakthrough treatments, including advanced therapies like CAR-T cell innovations21.
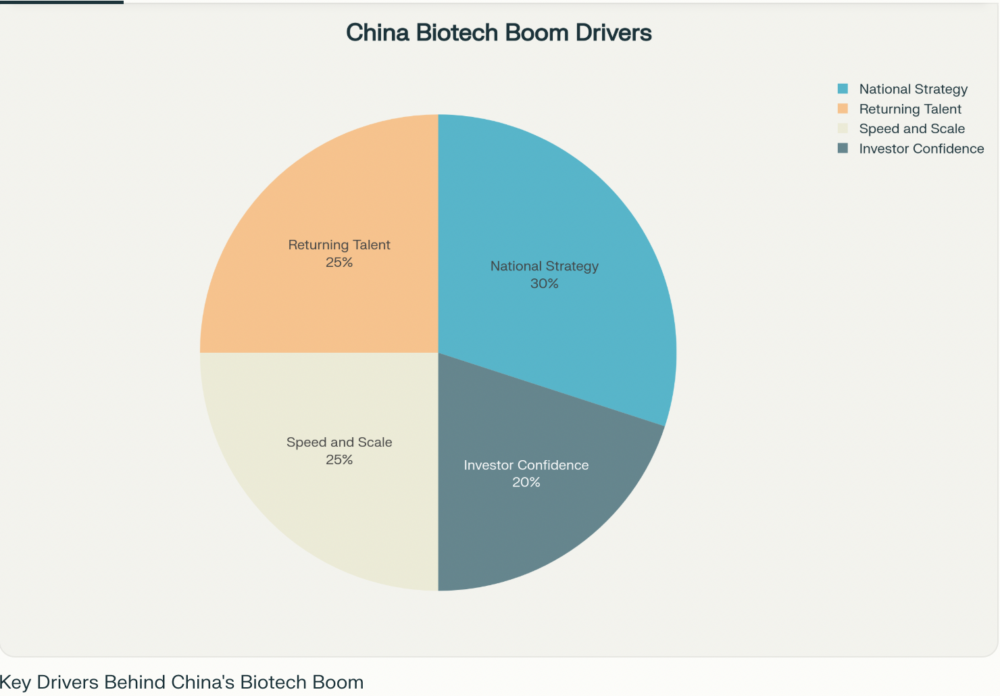
The Drivers Behind China’s Biotech Boom
- National Strategy: Policies like “Made in China 2025” propelled biotechnology to the top of the country’s priorities, pouring investment into research, infrastructure, and cross-border partnerships12.
- Returning Talent: A wave of scientists trained in the U.S. and Europe are returning to Chinese labs, bringing expertise and a drive to innovate at home3.
- Speed and Scale: More flexible regulations and abundant funding have enabled Chinese companies to move therapies from concept to clinical trial at lightning pace, outstripping their Western peers45.
Winning on the World Stage
- Global Recognition: Chinese drugmakers are rapidly closing the gap with U.S. and European giants. They’re not just producing for the local market—homegrown therapies are winning approval abroad and attracting blockbuster licensing deals with global players like Pfizer and Bristol-Myers Squibb67.
- Investor Confidence: International investors and Big Pharma are pouring billions into China’s biotech firms, eager to tap into the rich R&D pipeline and innovative platforms emerging from cities like Shanghai and Shenzhen63.
- Cost Advantage: China’s ability to develop drugs faster and often more cheaply is setting new benchmarks, making waves in global pricing and market access42.
Challenges and Competitive Tensions
- Regulatory Headwinds: As the West wakes up to China’s new biotech power, some U.S. policymakers are tightening scrutiny and investment restrictions, hoping to protect sensitive technology and maintain an edge74.
- Quality and Commercialization: Although China leads in trials and pipeline growth, veteran observers note that Western companies still have an advantage in successfully commercializing groundbreaking drugs on a global scale5.
- Enduring Rivalry: This biotechnology rivalry now sits alongside those in AI and electric vehicles—shaping new superpower dynamics in global health13.
The Takeaway
China’s advance in biotechnology is more than a local success story—it’s a paradigm shift that’s stirring up the global pharmaceutical ecosystem. As Chinese drugmakers sprint ahead, Western Big Pharma faces its most formidable challenger yet.

MedicinManPR with SlamDunk Creatives
- https://www.bloomberg.com/news/features/2025-07-13/china-drugmakers-catching-up-to-us-big-pharma-with-new-medicine-innovation
- https://www.biotecnika.org/2025/07/beyond-the-wall-chinas-biotech-innovation-drug-development/
- https://medicalbuyer.co.in/biotech-industry-experiencing-tectonic-shift-driven-by-chinese-drugmakers/
- https://www.cnbc.com/2025/06/05/chinas-quickly-gaining-an-edge-over-the-us-in-biotech.html
- https://itif.org/publications/2024/07/30/china-is-no-longer-a-biotech-imitator/
- https://www.bloomberg.com/opinion/articles/2025-06-10/why-china-biotech-is-getting-its-own-deepseek-moment
- https://informaconnect.com/uploads/Sourcing-Biotech-Innovation-in-China-ChinaBio-Special-Report2025-bc8b69062586ed2e3c90ab4a4aa05956.pdf
- https://ppl-ai-file-upload.s3.amazonaws.com/web/direct-files/attachments/12063389/7202433e-0265-42d0-b928-6f4a67de4871/CHS_Employee-Service-Rule-Book_SP_20.05.2025.docx
- https://www.bloomberg.com/news/articles/2025-06-15/china-s-biotech-moment-ignites-a-60-stock-rally-that-beats-ai
- https://www.archyde.com/china-biotech-breakthrough-reshapes-global-drug-development/
- https://www.axios.com/2025/05/29/china-biotech-boom-us-drug-trials
- http://epaper.chinadaily.com.cn/a/202405/31/WS6659163da310df4030f51ef5.html
- https://www.fdd.org/analysis/2025/06/03/china-races-ahead-in-biotech-trials-is-the-u-s-stuck-in-second-place/
- https://oncodaily.com/blog/norman-ng-329751
- https://www.bloomberg.com/news/videos/2025-07-15/china-biotech-s-stunning-advance-challenges-the-west
- https://www.business-standard.com/world-news/china-s-biotech-surge-shifts-global-drug-innovation-stirs-us-tension-125071400104_1.html
- https://www.chinadaily.com.cn/a/202406/04/WS665e7fe7a31082fc043cacb6.html
- https://www.bloomberg.com/news/articles/2025-03-10/china-s-biopharma-and-biotech-sector-is-booming
- https://www.biopharmadive.com/news/biotech-us-china-competition-drug-deals/737543/
- https://servier.com/en/newsroom/china-global-pharmaceutical-innovation/
- https://rhg.com/research/was-made-in-china-2025-successful/