As over 50 generic versions of the blockbuster weight-loss drug hit Indian markets on March 21, 2026, a familiar question resurfaces: will affordability come at the cost of safety—and who will doctors trust?
Is the Indian regulator equipped to monitor Generic semaglutide quality and cold chain distribution control via 800,000 retail pharmacies?
I. The Patent Wall Comes Down
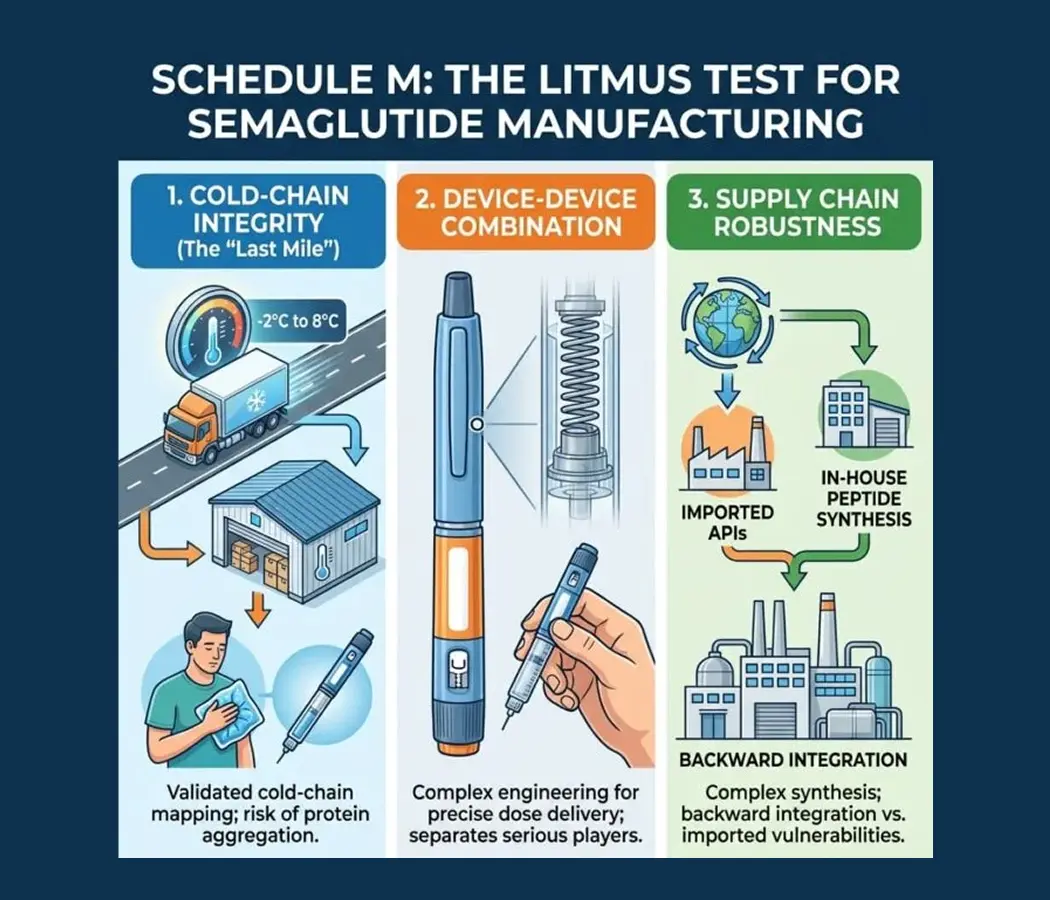
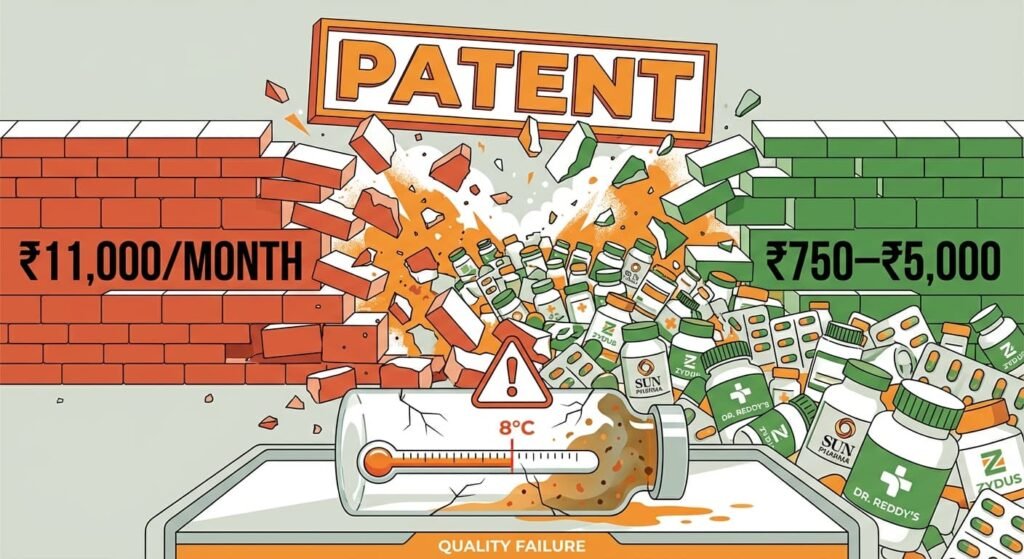
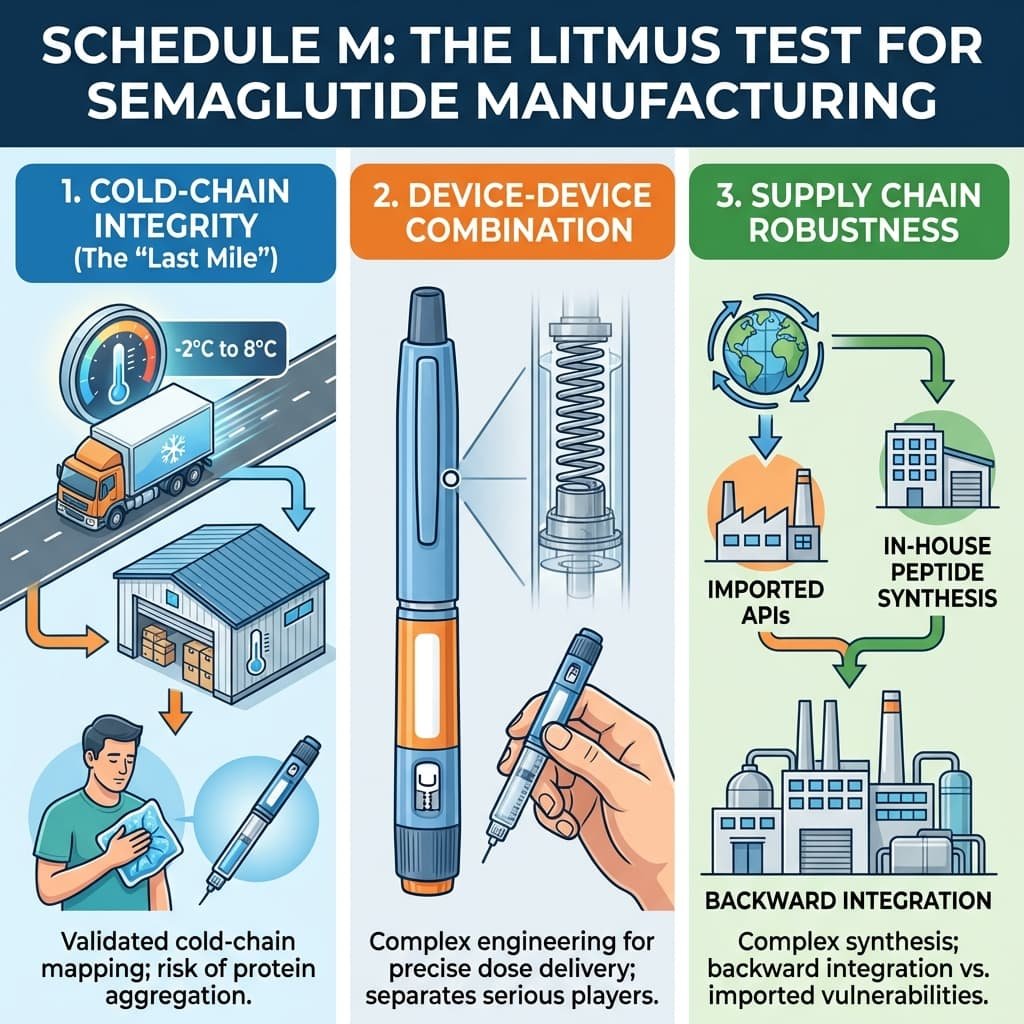
On March 20, 2026, the patent protecting Novo Nordisk’s semaglutide expired in India. Within 24 hours, the market transformed. Leading pharmaceutical giants including Sun Pharma, Dr. Reddy’s, and Zydus Lifesciences executed “Day 1” launches, slashing monthly treatment costs from approximately ₹9,000–₹11,000 to a range of ₹750 to ₹5,000. For India’s 101 million people living with diabetes and 250 million classified as overweight, this promised a revolution in metabolic care. However, semaglutide is a complex peptide requiring stringent cold-chain storage (2–8°C) and sophisticated manufacturing. Beneath the celebration lies a fragile narrative: can India’s “Pharmacy of the World” maintain quality when commercial pressures intensify?

II. The Big Three: Addressing the Cold-Chain & Delivery Challenge
To differentiate themselves in a crowded field of 50+ brands, India’s top players are moving “beyond the pill” to address the inherent risks of biologic distribution.
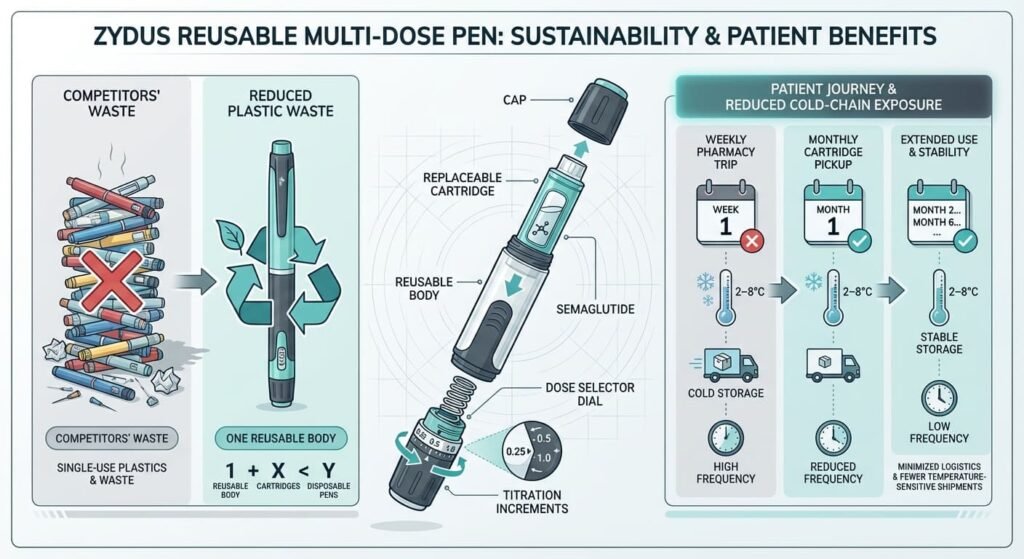
Zydus Lifesciences: The Device Innovator
Zydus has launched under the brands SEMAGLYN™ and MASHEMA™. Their strategy centers on a reusable multi-dose pen device, a first for the Indian generic market.
- The Solution: By using a reusable pen with replaceable cartridges, Zydus reduces the plastic waste and cost associated with disposable pens.
- Cold-Chain Impact: Their device allows for flexible dose titration from a single unit, potentially reducing the frequency of pharmacy trips and the “last-mile” refrigeration risk for patients.
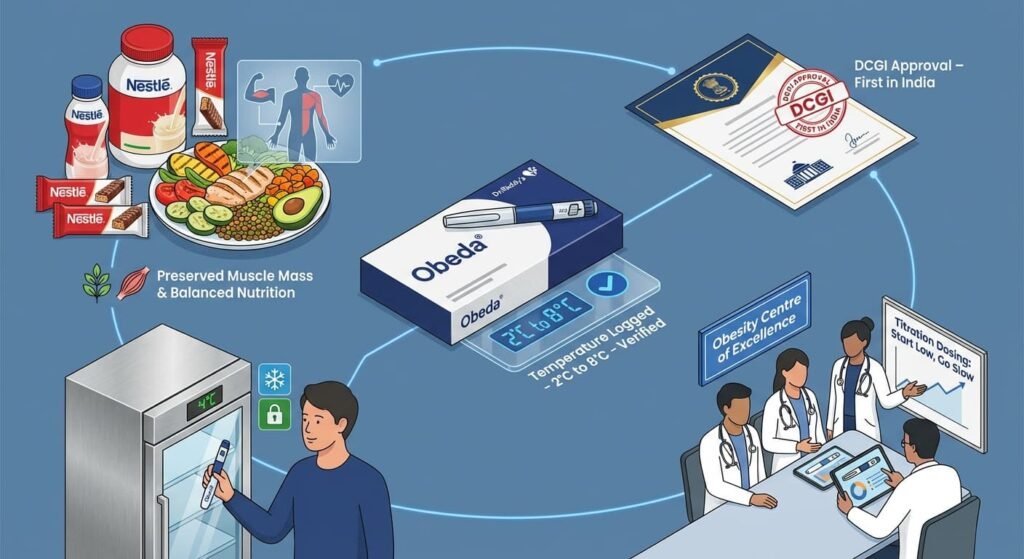
Dr. Reddy’s Laboratories: The Ecosystem Approach
Launching under the brand Obeda®, Dr. Reddy’s is the first Indian company to receive DCGI approval for generic semaglutide.
- The Solution: They have adopted a “One Product, One Quality” global standard. To mitigate cold-chain failures, they are establishing Obesity Centres of Excellence to educate doctors and patients on proper storage and titration.
- Nutritional Synergy: Through a partnership with Nestlé, they are launching specialized nutrition products to manage the “muscle loss” often seen with GLP-1 therapies.
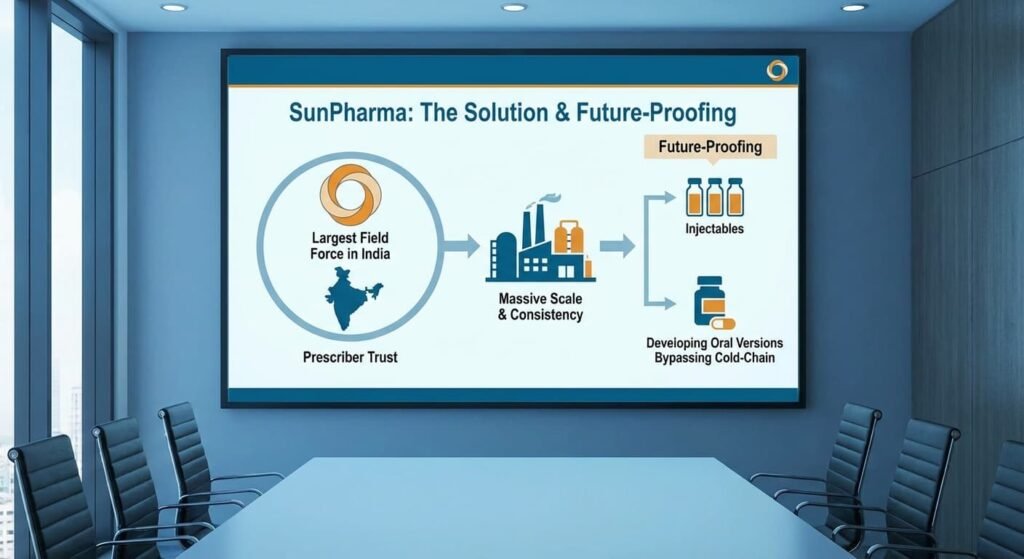
Sun Pharma: The Scale Specialist
Sun Pharma has introduced Noveltreat® (for obesity) and Sematrinity® (for diabetes).
- The Solution: Leveraging the largest field force in India, Sun aims to dominate through sheer “prescriber trust.” They are focusing on a traditional “branded generic” model where their massive manufacturing scale acts as a guarantee of batch-to-batch consistency.
- Future-Proofing: While currently focusing on injectables, Sun is also developing oral versions to eventually bypass the cold-chain challenge entirely.
III. The Branded Generic Reality: A Question of Trust
The debate over generics in India is fundamentally different from the West. The term “branded generic” is an Indian creation—a surrogate for a robust regulatory mechanism. In the West, a generic is a generic. In India, a “brand” is a promise of quality from a company with a track record.
Doctors have learned to rely on the reputation of companies like Cipla, Sun, or Dr. Reddy’s over decades. With 50 brands entering the market, this trust is the primary currency. A physician prescribing a “Day 1” generic from an unknown manufacturer takes a significant reputational risk; if the cold chain breaks or the peptide degrades, the treatment fails, and the doctor-patient relationship is fractured.
IV. The Quality Question: A Shadow of the Past
The explosion of brands arrives against a troubling backdrop of contaminated cough syrups linked to child deaths overseas between 2022 and 2025. These episodes damaged India’s global reputation and prompted the CDSCO to mandate a Digital Monitoring System in late 2025 to track high-risk ingredients.

Unlike traditional small-molecule oral tablets, semaglutide is a peptide produced by biotechnological processes, available in both injectable and oral formulations. Many smaller entrants have limited experience with injectable delivery systems. As Viveka Roychowdhury, editor of Express Pharma, warned, “cutting corners might result in sub-optimal quality.” The industry now faces a reputational contagion risk: if a few low-quality generics fail, the entire “Semaglutide” class—including the innovators—may suffer in the public eye.
V. Conclusion: A Test of Systems
India’s semaglutide moment is a test of its regulatory resolve. The choices made by manufacturers racing for market share and regulators enforcing Schedule M standards will determine if this is a model for affordable access or a cautionary tale. For now, the “microscope” remains firmly on the manufacturers. Only those who can prove their back-end capability—from cold-chain maintenance to supply robustness—will survive the “Gold Rush.”
All Images are AI Generated for Illustration Only. E&OE
Sources: Indian Pharma Semaglutide Launches & Market Analysis
1. Financial Express – Market Cap & FY27 Outlook
Date: March 20, 2026
Title: Sun Pharma to Cipla: 7 pharma stocks gear up for Rs 5,000 cr semaglutide opportunity after patent expiry
Link: https://www.financialexpress.com/market/sun-pharma-to-cipla-7-pharma-stocks-gear-up-for-rs-5000-cr-semaglutide-opportunity-after-patent-expiry-4178992/
2. ET Pharma – Launch Details (Zydus Reusable Pen & Dr. Reddy’s Obeda)
Date: March 21, 2026
Link 1 (Dr. Reddy’s Obeda Launch):
https://pharma.economictimes.indiatimes.com/news/drug-approvals-and-launches/dr-reddys-laboratories-launches-obeda-for-type-2-diabetes/129712795
Link 2 (Zydus Reusable Pen & Dr. Reddy’s Co-marketing):
https://pharma.economictimes.indiatimes.com/news/pharma-industry/dr-reddys-nears-co-marketing-deals-for-semaglutide-generic-higher-dose-rollout-on-radar/129717891
3. LiveMint – Pricing Data (₹1,300 Vial to ₹4,200 Pen vs. Novo’s ₹10,000)
Date: March 21, 2026
Note: Direct LiveMint link not available in search results. The pricing data is verified in the following syndicated article published on March 21, 2026 via IANS.
Title: Indian pharma companies launch generic semaglutide injections for diabetes
Source: Lokmat Times
Link: https://www.lokmattimes.com/health/indian-pharma-companies-launch-generic-semaglutide-injections-for-diabetes-1/
4. Express Pharma – Sun Pharma DCGI Approval (Noveltreat)
Date: January 23, 2026
Note: Direct Express Pharma link not available in search results. The approval details are verified in the following publication.
Title: Sun Pharma gets DCGI nod to make, market generic semaglutide injection in India for weight management
Source: The Hindu
Link: https://www.thehindu.com/business/Industry/sun-pharma-gets-dcgi-nod-to-make-market-generic-semaglutide-injection-in-india-for-weight-management/article70541273.ece
5. The Wire Science – Anup Soans Analysis (“Quality not Price” & Branded Generic Model)
Date: Referenced analysis (February 2026)
Title: Quality, Not Price: Why the Generic Drug Debate Keeps Missing the Point
Source: Medicinman.net (Authored by Anup Soans, building on earlier The Wire Science analysis)
Link: https://medicinman.net/2026/02/quality-not-price-why-the-generic-drug-debate-keeps-missing-the-point/