
Abstract
When Intas Pharmaceuticals launched Razumab in June 2015 as the world’s first biosimilar of ranibizumab, it promised to transform retinal disease treatment in India through affordability. A decade later, the anti-VEGF market has been fundamentally altered, with multiple biosimilars challenging multinational innovator dominance. This article examines Razumab’s trajectory through three phases: regulatory navigation, clinical validation under scrutiny, and market maturation. It analyzes the clinical evidence that established biosimilar equivalence, critically evaluates early safety concerns that tested the product’s viability, and assesses the evolving regulatory landscape that will shape the next generation of Indian biosimilars.

Introduction
In June 2015, Intas Pharmaceuticals announced the launch of Razumab, describing it as the world’s first biosimilar of ranibizumab . The company priced the product approximately 25% lower than Novartis’s Lucentis—₹25,000 versus ₹35,000 per vial—with the stated objective of making treatment accessible to a broader patient population in a country estimated to have 15–20 million cases of retinal diseases.
The launch represented more than a commercial milestone. It was a test case for India’s ability to develop complex biologic products that could compete with multinational innovators while maintaining safety standards. The intervening decade has seen Razumab’s sales grow from approximately 2,800 vials in 2015 to over 120,000 by 2020, alongside the entry of additional biosimilars including Enzene’s version. Yet this growth was neither linear nor assured.
This article assesses how Razumab altered the treatment landscape, analyzes the clinical data that addressed initial skepticism, and evaluates the safety challenges that tested the product’s viability during its early years.

Part I: The Regulatory Framework for Biosimilar Approval
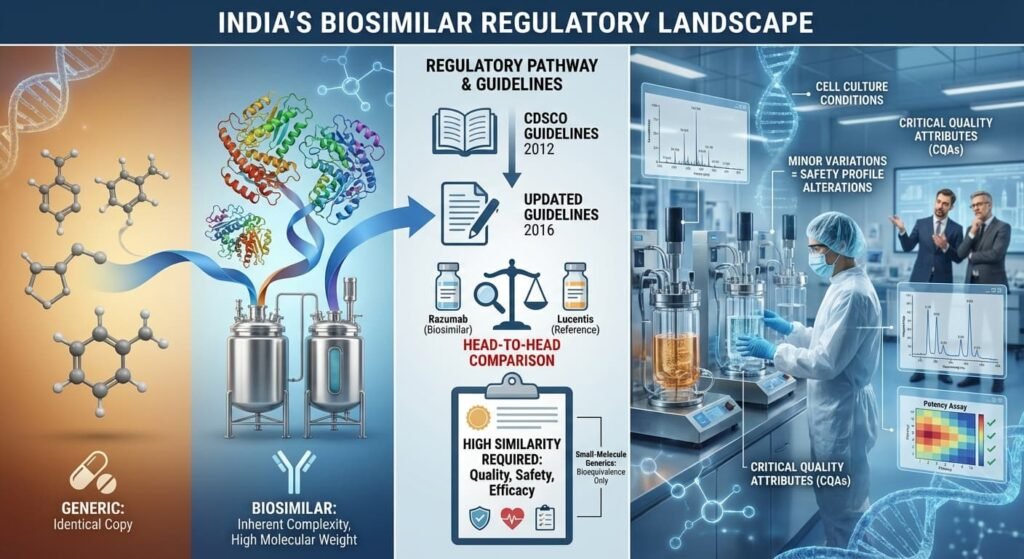
Understanding Razumab’s trajectory requires familiarity with biosimilar approval pathways. Unlike generic drugs—which are chemically identical copies—biosimilars are derived from living cells and possess inherent complexity, including high molecular weight and sensitivity to manufacturing conditions.
When Intas developed Razumab in-house, it faced India’s biosimilar guidelines (established in 2012 and updated in 2016), which mandated extensive head-to-head comparisons with the reference biologic. The Central Drugs Standard Control Organisation (CDSCO) required evidence demonstrating “high similarity” in quality, safety, and efficacy—a more rigorous standard than the bioequivalence requirements for small-molecule generics.
The analytical challenge was significant: even minor variations in manufacturing processes—including cell culture conditions—could alter a biosimilar’s safety profile. Regulators expected orthogonal analytical techniques to assess Critical Quality Attributes (CQAs), and initial skepticism from multinational competitors often centered on whether Indian manufacturers could maintain this consistency.
Part II: Early Market Entry and Safety Concerns
Razumab’s launch was accompanied by immediate market impact but also by safety signals that threatened its viability. Within two months of commercial availability, reports emerged of adverse reactions, including intraocular inflammation in patients receiving the injection .
The 2015–2017 Safety Events
In August 2015, Intas curtailed distribution of Razumab following reports of inflammation in approximately nine patients. The Vitreo Retina Society of India (VRSI) issued a cautionary communication to its members about adverse reactions. Intas attributed the incidents to cold-chain maintenance issues in its distribution network and announced measures to strengthen temperature monitoring with data loggers for each delivery .
A second, more significant safety event occurred in 2017. The VRSI reported that 11 eyes across five centers experienced inflammation after receiving injections from a specific batch (number 18020020). Intas recalled all vials from this batch—182 of which had been administered to patients—for internal quality control testing. The company advised clinicians to avoid using that batch while investigations proceeded .

For a product administered intravitreally (injected directly into the eye), safety concerns of this nature could have permanently derailed market acceptance. The company’s response—recalling the batch, implementing enhanced cold-chain protocols, and committing to transparency with the ophthalmology community—proved critical to maintaining clinician confidence.
Intas noted in its defense that published data for the innovator ranibizumab reported ocular inflammation in approximately 18% of patients, and that the incidence in its product remained within comparable limits . While this did not eliminate concern, it contextualized the events within known class effects.
Part III: Clinical Validation—Building the Evidence Base
The turning point for Razumab came through the accumulation of real-world evidence demonstrating comparable efficacy and safety to the innovator product.
The RE-ENACT Studies
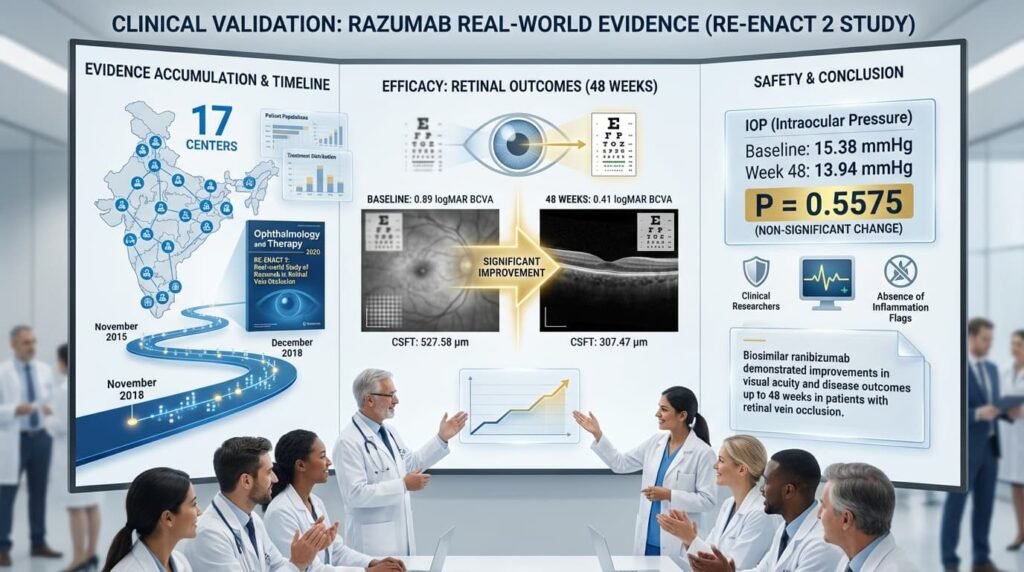
The RE-ENACT 2 study, published in Ophthalmology and Therapy in 2020, analyzed data from patients with retinal vein occlusion (RVO) treated with Razumab across 17 centers between November 2015 and December 2018. The findings showed significant improvements in clinical outcomes:
- Visual acuity: Best Corrected Visual Acuity (BCVA) improved from a baseline of 0.89 logMAR to 0.41 logMAR at 48 weeks
- Anatomical outcomes: Central subfield thickness (CSFT) decreased from 527.58 µm to 307.47 µm
- Safety: Intraocular pressure changes were non-significant (15.38 mmHg at baseline versus 13.94 mmHg at week 48; P = 0.5575)
The study concluded that biosimilar ranibizumab “demonstrated improvements in visual acuity and disease outcomes up to 48 weeks in patients with RVO without any new safety concerns”.
Head-to-Head Comparison with Innovator Ranibizumab
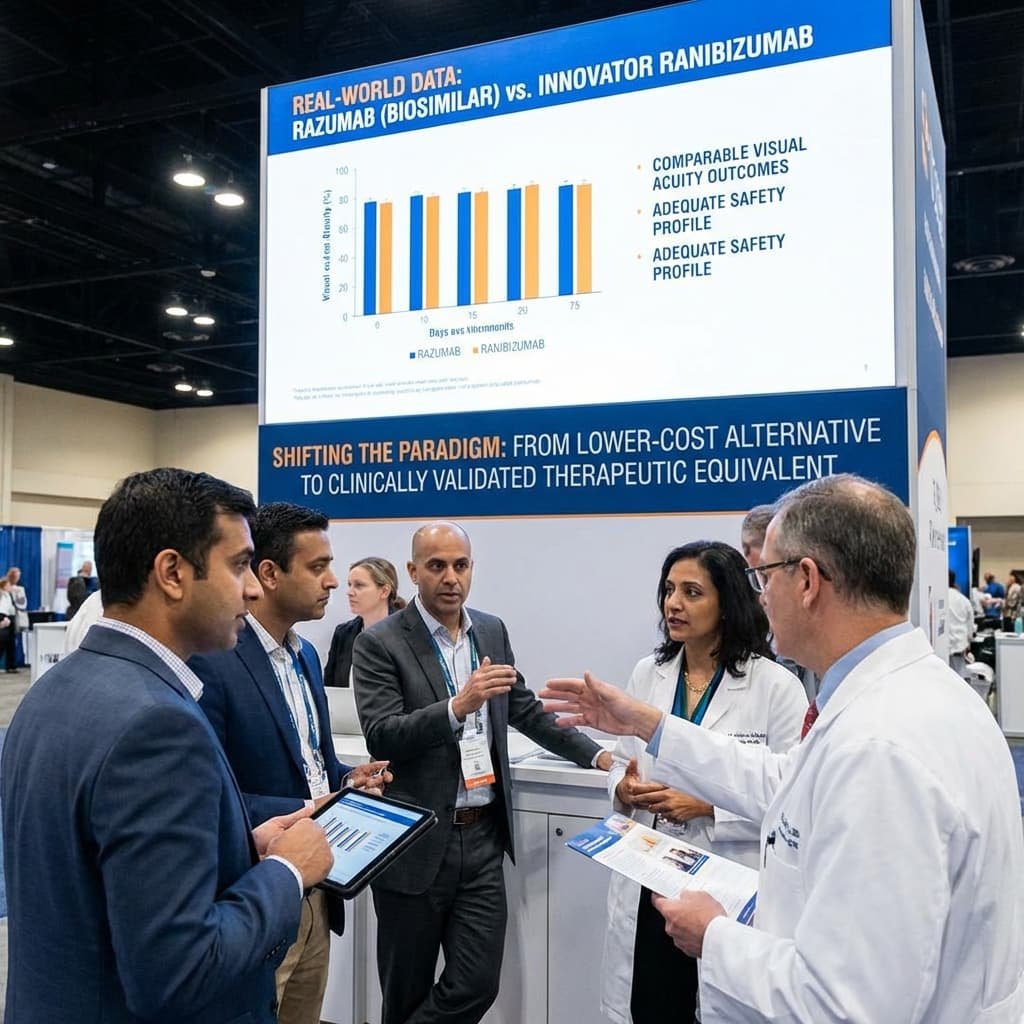
A landmark comparative study published in Ophthalmology and Therapy in 2022 directly evaluated Razumab against Lucentis in patients with polypoidal choroidal vasculopathy (PCV)—a condition particularly prevalent in Asian populations .
The study analyzed 22 eyes receiving innovator ranibizumab and 19 eyes receiving Razumab. Notable findings included:
| Outcome Measure | Innovator (IRm) | Biosimilar (BRm) | Statistical Significance |
|---|---|---|---|
| Visual acuity gain at 52 weeks | Significant improvement (P = 0.001) | Comparable gain (P = 0.19 between groups) | P = 0.19 |
| Subretinal fluid resolution | Comparable | Comparable | P = 0.8 |
| Intraretinal fluid resolution | Comparable | Comparable | P = 0.47 |
| Safety events | None | One case of mild anterior uveitis (5.26%) | N/A |
The authors concluded: “Our real-world data demonstrated the ranibizumab biosimilar Razumab to have comparable visual acuity outcomes to the innovator ranibizumab molecule with an adequate safety profile in the management of PCV” .
This evidence proved decisive. It shifted Razumab’s market positioning from a lower-cost alternative to a clinically validated therapeutic equivalent—a distinction essential for sustained adoption among risk-averse clinicians.
Part IV: The Competitive and Intellectual Property Landscape
The narrative that Razumab succeeded against “MNC headwinds” requires nuance. Public records do not indicate that Novartis pursued patent litigation against Razumab immediately post-launch, though the broader intellectual property environment for biosimilars in India remained contentious during this period. In 2013, Novartis had lost a landmark Supreme Court case regarding the patenting of Glivec (imatinib) under Section 3(d) of the Patents Act, which prohibits “evergreening” of patents. This decision, along with broader industry concerns about India’s intellectual property regime, created a challenging environment for multinational pharmaceutical companies operating in the country .
However, the safety events of 2015–2017 validated legitimate concerns about whether biosimilar manufacturing could consistently meet the quality standards required for intravitreal administration. The “headwinds” Razumab faced were not solely commercial in nature—they reflected genuine scientific and clinical questions about manufacturing consistency and product safety. That Intas navigated these challenges, strengthened its quality systems, and produced robust clinical data is a testament to the company’s resilience, but it also underscores that initial skepticism was not without foundation.

Part V: Regulatory Evolution—2025 Guidelines and Beyond
By 2026, the regulatory landscape for biosimilars in India has evolved substantially. In May 2025, CDSCO published draft revised guidelines that represent “a progressive shift toward harmonizing Indian regulatory requirements with global standards set by the EMA and FDA” .
Key changes in the 2025 draft guidelines include:
- De-emphasis on animal testing: The guidelines encourage in vitro assays over in vivo animal studies, following the global 3Rs principle (Replace, Reduce, Refine)
- Potential waiver of efficacy trials: Under specific conditions, confirmatory Phase 3 efficacy trials may be waived if pharmacokinetic/pharmacodynamic equivalence is demonstrated with robust biomarkers
- Analytics-first approach: Regulators now emphasize “next generation analytical methodologies” and orthogonal techniques to assess Critical Quality Attributes
These changes reflect international convergence in biosimilar regulation and are likely to accelerate future product development while maintaining rigorous standards for analytical similarity.

Competitive Landscape and Market Evolution
Intas no longer holds exclusive position in the ranibizumab biosimilar market. Enzene Biosciences launched its version in 2023, and additional competitors are expected as the market matures. The anti-VEGF segment has transitioned from a “launch-centric phase” to a multi-therapy growth engine where price is no longer the sole differentiator—clinical experience, supply chain reliability, and long-term safety track records increasingly determine prescribing patterns.
Conclusion
Razumab’s decade-long journey illustrates both the potential and the challenges inherent in biosimilar development. The product began with an ambitious pricing strategy, encountered early safety concerns that tested its viability, validated its clinical profile through rigorous real-world evidence, and ultimately achieved acceptance as a therapeutic equivalent to the innovator product.
Several lessons emerge for the next generation of Indian biosimilars:
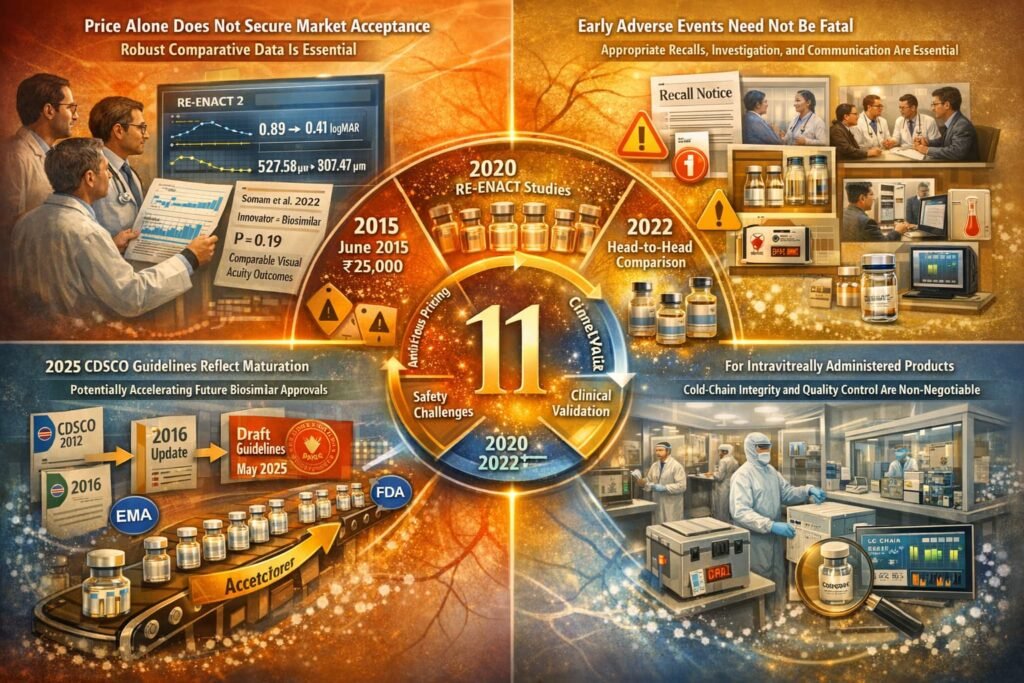
- Clinical evidence matters: Price alone does not secure market acceptance; robust comparative data is essential.
- Safety transparency is critical: Early adverse events need not be fatal if managed with appropriate recalls, investigation, and communication.
- Regulatory alignment facilitates development: The 2025 CDSCO guidelines reflect maturation of India’s regulatory framework, potentially accelerating future biosimilar approvals.
- Manufacturing consistency underpins credibility: For intravitreally administered products, cold-chain integrity and quality control are non-negotiable.
As India’s biosimilar sector moves into its next decade, Razumab’s legacy is not simply that it was first. It is that it survived the convergence of commercial, clinical, and regulatory challenges to demonstrate that affordable biosimilars can achieve safety and efficacy standards comparable to multinational innovator products.
References
- Launch and pricing (2015) – PhIRDA. (2015). Novartis’ $4 billion ophthalmology drug Lucentis faces first biosimilar challenge as India’s Intas launches Razumab. China Pharmaceutical Innovation Promotion Association.
- Safety recall (2017) – The Indian Express. (2017, April 3). Patients report eye inflammation, Intas recalls its drug batch.
- RE-ENACT 2 study (RVO) – Sharma, S., RE-ENACT 2 Study Investigators Group, et al. (2020). A Multicenter, Retrospective Study (RE-ENACT 2) on Razumab™ in Retinal Vein Occlusion. Ophthalmology and Therapy, 9(3), 625–639.
- Head-to-head comparison (PCV) – Soman, M., Nair, I., Sheth, J. U., & Nair, U. (2022). Innovator Versus Biosimilar Ranibizumab in Polypoidal Choroidal Vasculopathy: Real-World Evidence. Ophthalmology and Therapy, 11(3), 1175–1186.
- CDSCO 2025 draft guidelines – Pearce IP. (2025, May 6). India’s CDSCO Publishes Draft Revised Biosimilar Guidelines for Stakeholder Comment.
- CDSCO 2025 guidelines (Lexology) – Lexology. (2025, May 6). India’s CDSCO Publishes Draft Revised Biosimilar Guidelines for Stakeholder Comment.
- Early safety concerns (2015) – The Economic Times. (2015, August 21). Intas cuts Razumab supplies after adverse eye reactions.
- Launch announcement (2015) – Medthority. (2015, June 20). Razumab (ranibizumab biosimilar) is launched in India for degenerative conditions of the eye.
Disclosure Statement
The author has no financial or proprietary interest in any product or company mentioned in this article. No funding was received for the preparation of this manuscript.
Suggested Citation:
Anup Soans. Razumab at 11: A Decade of Disruption, Safety Lessons, and the New Biosimilar Reality. MedicinMan. March 2026.

All Images are AI Generated for Illustration Only. E&OE






