
Walk into almost any clinic today and you will notice a familiar pattern in prescriptions. Alongside conventional medicines, doctors frequently recommend products described as “nutritional support” for joints, bones, metabolism, heart health, immunity, or gut function.
These products—collectively known as nutraceuticals—promise to strengthen the body, nourish tissues, and sometimes even slow the progression of chronic disease.
Yet nutraceuticals occupy an unusual and contested space in healthcare. They are not conventional drugs, yet they are often prescribed like medicines. They are not merely foods, yet they carry claims of therapeutic benefit.
In many ways, nutraceuticals exist at the intersection where medicine, marketing, and patient psychology meet. Understanding their role requires a critical look at the evidence, the motivations of those who prescribe and consume them, and the regulatory landscape that allows them to flourish.

The Therapeutic Gap That Created the Market
The rapid growth of nutraceuticals is not accidental. It reflects the structure of modern medicine itself.
Many chronic diseases evolve slowly over years, during which treatment options may be limited.
Consider Osteoarthritis, one of the most common causes of disability worldwide. Patients are usually advised physiotherapy, lifestyle modifications, and pain relief medications. Surgery is reserved for advanced disease.
But between early symptoms and surgical intervention lies a long period—often lasting years—during which patients seek something that might protect or repair their joints.

Similar gaps exist in other conditions:
· Osteoporosis, where long-term bone support is needed
· Type 2 Diabetes, where metabolic support supplements are often used
· Hyperlipidemia, where patients seek natural lipid-lowering options
· Irritable Bowel Syndrome, where microbiome therapies are increasingly explored
Nutraceuticals have flourished in these therapeutic grey zones, offering patients and physicians a supportive middle path between lifestyle changes and pharmaceuticals. However, while nutraceuticals have flourished in these gaps, the scientific evidence supporting their use varies dramatically—from robust clinical data for some indications to plausible but unproven theories for others.

The Expanding Portfolio of Nutraceuticals: A Critical Look at the Evidence
Over the past two decades, nutraceuticals have diversified into several major therapy areas. A scientifically robust view requires examining not just what is marketed, but what the highest quality evidence demonstrates.
Joint and Musculoskeletal Health
This remains one of the largest categories. Products frequently contain Glucosamine, Chondroitin sulfate, collagen peptides, and micronutrients believed to support cartilage and connective tissue.
The Evidence: Large-scale, high-quality studies and meta-analyses, including those published in the BMJ and the Cochrane Database of Systematic Reviews, have shown mixed results. While some patients report symptomatic relief, the evidence suggests that these supplements are unlikely to repair cartilage or modify the structure of the disease. Any pain relief is often modest and, in the most rigorous trials, can be comparable to a placebo.
Bone Health
Bone formulations commonly include Calcium citrate and Vitamin D, which are widely recommended to maintain bone density and reduce fracture risk.
The Evidence: The evidence for calcium and vitamin D in preventing fractures in older adults is well-established, particularly in institutionalized individuals with known deficiencies. However, recent large trials have questioned the universal benefit for community-dwelling adults, suggesting the greatest impact is in those with confirmed low nutrient levels.
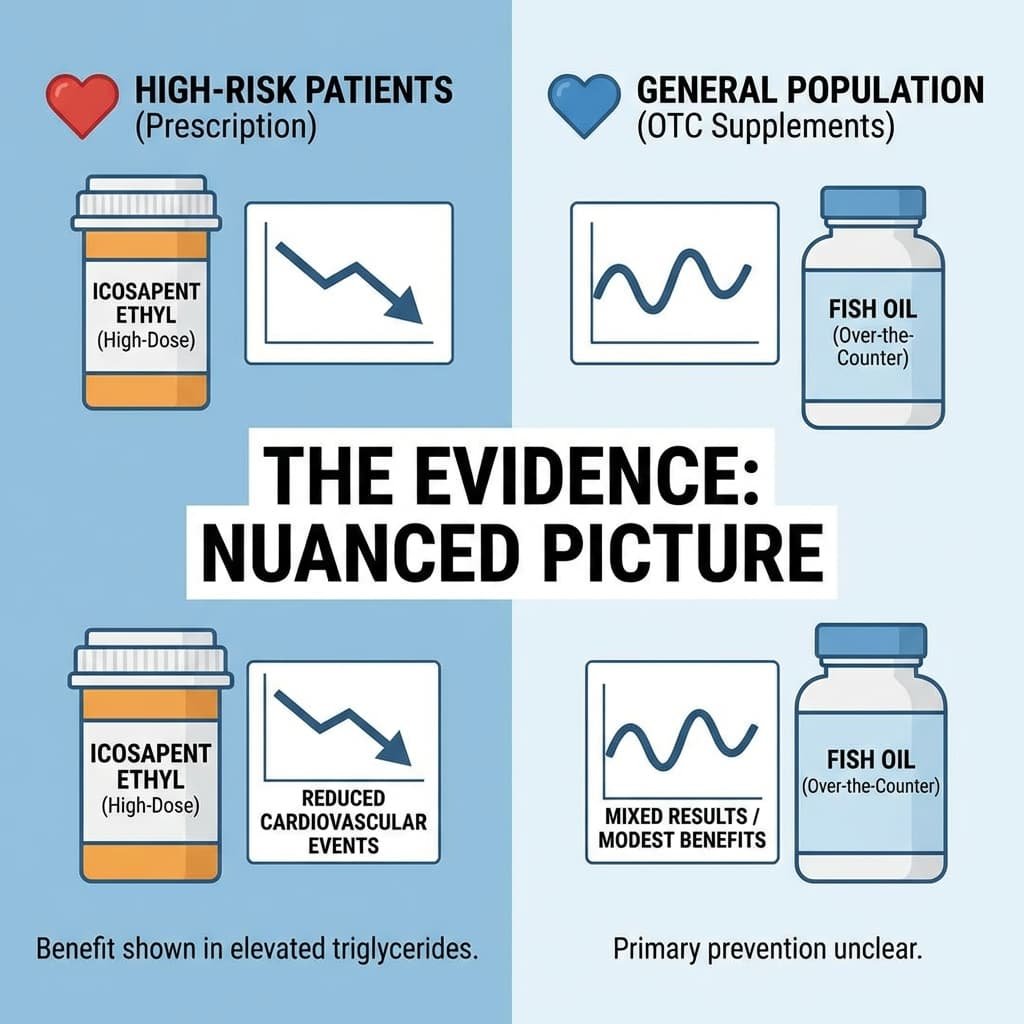
Cardiovascular Health
Supplements containing Omega-3 fatty acids (fish oil) are often promoted for heart health and lipid management.
The Evidence: Here, the picture is more nuanced. High-dose prescription omega-3s (like icosapent ethyl) have shown benefit in reducing cardiovascular events in high-risk patients with elevated triglycerides. However, over-the-counter fish oil supplements for primary prevention in the general population have yielded more mixed results in recent large trials, with benefits appearing modest at best.
Metabolic Health
Compounds such as Alpha-lipoic acid are sometimes used as adjunct therapies for metabolic disorders and diabetic neuropathy.
The Evidence: Some studies suggest alpha-lipoic acid may improve insulin sensitivity and reduce symptoms of diabetic neuropathy. However, trials are often small and of short duration, and the optimal dosing and long-term effects remain areas of ongoing investigation.
Gut and Microbiome Health
Digestive health supplements frequently rely on Probiotics, intended to support a balanced intestinal microbiome.
The Evidence: The evidence for probiotics is highly strain-specific. While certain strains have demonstrated efficacy for specific conditions like antibiotic-associated diarrhea or managing symptoms of irritable bowel syndrome, generalized claims of improved “gut health” often outpace the scientific data.
Across these therapy areas, the scientific evidence ranges from well-established to emerging and, in some of the most popular categories, surprisingly inconclusive. Yet demand continues to grow globally.

The Psychology of Chronic Disease
One reason nutraceuticals resonate strongly with patients lies in the psychology of chronic illness.
People living with degenerative diseases often feel that their bodies are gradually deteriorating. Conventional medicines may relieve symptoms but rarely promise restoration.
Nutraceuticals, however, are frequently framed around a different narrative: support, repair, rebuild, strengthen.
This narrative is powerful. It suggests that the body can still heal itself—provided it receives the right nutritional support. This appeal to a patient’s desire for agency and control is a potent psychological force.
This powerful narrative can, in itself, have therapeutic value. The ‘meaning response’ or placebo effect—whereby a patient’s belief in a treatment leads to real improvements in symptoms—is a well-documented phenomenon. For conditions where the nutraceutical’s specific biochemical effect may be modest, this psychological boost could account for a significant portion of the perceived benefit.

Why Physicians Continue to Prescribe Them
Doctors also face practical challenges when managing chronic disease.
Many pharmaceutical treatments are effective but carry risks when used long term. Anti-inflammatory drugs, for example, can cause gastrointestinal, renal, or cardiovascular side effects.
Nutraceuticals are often perceived as low-risk adjunct therapies and are generally well-tolerated. This perception, combined with the ‘pragmatic logic’ described below, makes them an easy recommendation. However, it’s crucial to note that ‘natural’ does not equal ‘risk-free.’ Potential for interactions with prescription drugs exists (e.g., St. John’s Wort, Vitamin K with warfarin), and due to less stringent regulation, the actual content and quality of supplements can vary significantly between brands.
In clinical practice, the reasoning often becomes pragmatic:
If a therapy might help and is unlikely to cause harm, it may be worth trying.
This practical logic has helped nutraceuticals become embedded in everyday medical practice.

The Role of Marketing
The nutraceutical market has also benefited from sophisticated branding and physician engagement.
Rather than positioning these products solely as retail supplements, many companies introduced them through medical representatives, clinical conferences, and continuing education programs.
This approach transformed nutraceuticals from consumer wellness products into doctor-endorsed therapies, reinforcing their credibility in clinical settings. Marketing materials often highlight biological plausibility and preliminary studies, while downplaying the inconclusive results from larger, more definitive trials.
The result is a category where scientific plausibility, patient expectations, and commercial strategy interact closely.

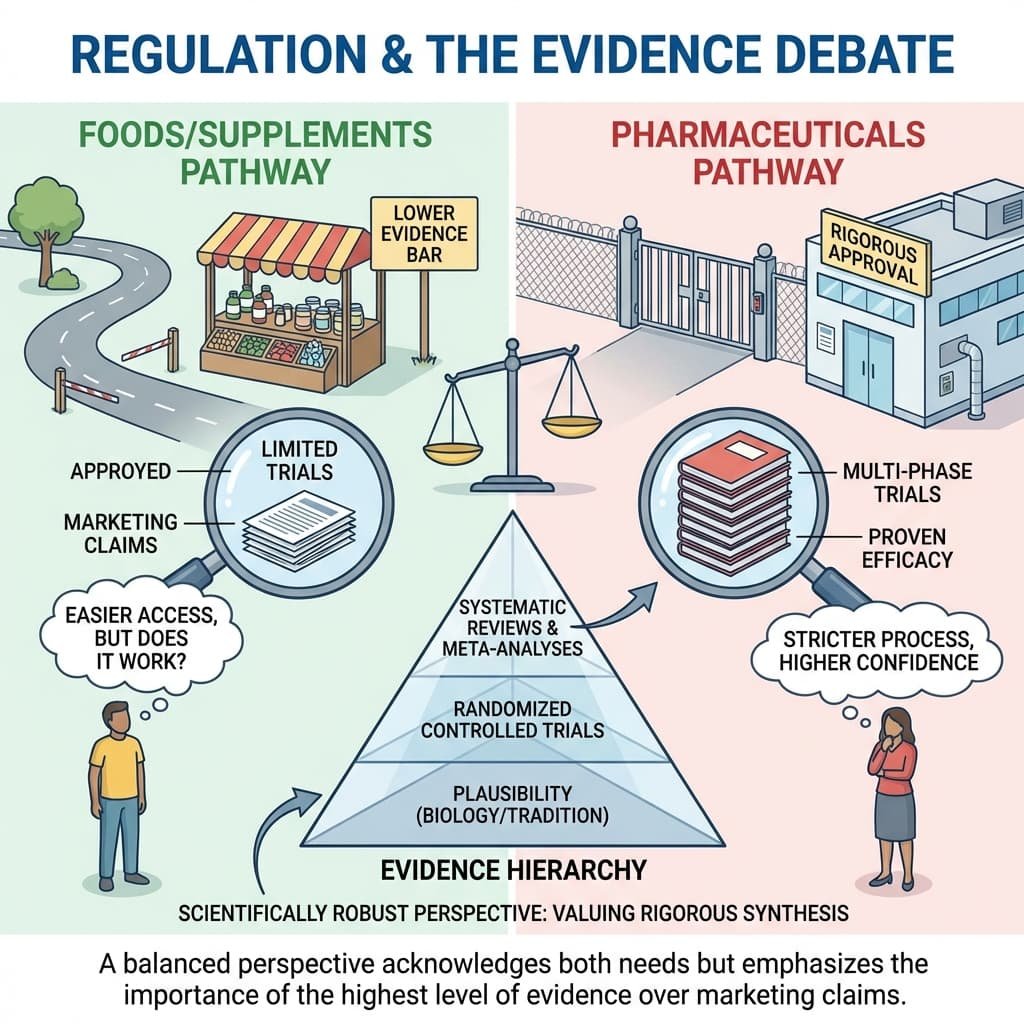
Regulation and the Evidence Debate
A continuing debate surrounds the regulatory status of nutraceuticals.
In many countries they are regulated as foods or dietary supplements rather than pharmaceuticals. As a result, the level of clinical evidence required for approval is often lower than for drugs. They do not need to prove efficacy through the same rigorous, multi-phase clinical trials required for new medications.
Critics argue that this lower regulatory bar allows products with limited evidence to proliferate, creating a market where marketing can outpace science. Supporters counter that these products fill a vital need in areas where pharmaceutical research is lacking.
A scientifically robust perspective would acknowledge both points but emphasize the importance of the evidence hierarchy. A product’s plausibility (based on biology or tradition) is the lowest form of evidence. It must be followed by rigorous randomized controlled trials and, most importantly, systematic reviews and meta-analyses that synthesize all available data. The key references in the appendix represent this highest level of evidence. Their conclusions are often more measured and less definitive than the marketing claims for the products they study.
The Next Frontier: From Supplements to Regenerative Medicine
Advances in regenerative medicine may eventually reshape the role of nutraceuticals in musculoskeletal care.
Emerging therapies such as Platelet-Rich Plasma therapy and Stem Cell Therapy aim to stimulate tissue repair more directly.
Although these technologies remain expensive and scientifically evolving, they represent a shift toward treatments that aim for true biological restoration rather than nutritional support alone.
It is more accurate to see this as a potential redefinition of the therapeutic ladder, rather than a simple replacement. In the future, patients might start with lifestyle changes, add nutraceuticals for general support, and then, if eligible and necessary, progress to more advanced (and expensive) regenerative biologic therapies for specific conditions.

Between Evidence, Marketing, and Hope
Nutraceuticals have grown rapidly because they address a reality of modern healthcare: chronic diseases often require long periods of management where treatment options are limited.
They offer patients something tangible to do, physicians something safe to recommend, and companies an opportunity to innovate within a relatively flexible regulatory environment.
In that sense, nutraceuticals are neither purely scientific breakthroughs nor merely marketing constructs. They are products of a healthcare ecosystem in which biology, belief, and business intersect.
And perhaps that is why they continue to flourish at the boundary where medicine meets marketing—and patient psychology. A scientifically literate approach neither dismisses them entirely nor accepts their claims at face value, but rather evaluates them with a critical eye on the evidence, an understanding of their psychological appeal, and a clear-eyed view of the market forces that propel them.

Appendix: Evidence Summary from Key References
The scientific evidence for commonly used nutraceuticals, particularly in musculoskeletal health, is often more nuanced than marketing claims suggest. The following high-quality systematic reviews and meta-analyses represent the highest level of evidence.
- Glucosamine and Chondroitin for Osteoarthritis
· Wandel S, Jüni P, Tendal B, et al. Effects of glucosamine, chondroitin, or placebo in patients with osteoarthritis of hip or knee: network meta-analysis. *BMJ. * 2010;341:c4675.
· Conclusion: This large network meta-analysis found that compared to placebo, neither glucosamine, chondroitin, nor their combination reduced joint pain or affected joint space narrowing in a clinically meaningful way. The authors concluded that health authorities and insurers should not cover these treatments, and they should not be used in clinical practice.
· Singh JA, Noorbaloochi S, MacDonald R, Maxwell LJ. Chondroitin for osteoarthritis. *Cochrane Database of Systematic Reviews. * 2015, Issue 1. Art. No.: CD005614.
· Conclusion: This Cochrane review concluded that compared to placebo, chondroitin did not improve pain or function in people with osteoarthritis in a clinically important manner. While it was safe, its benefit was minimal.
- Broader Reviews of Dietary Supplements
· Zeng C, Wei J, Li H, et al. Effectiveness and safety of Glucosamine, chondroitin, the two in combination, or celecoxib in the treatment of osteoarthritis of the knee. *British Journal of Sports Medicine. * 2018;52:1395-1402. Note: This reference is updated from the original prompt to a more recent, high-profile publication.
· Conclusion: This network meta-analysis found that while some dietary supplements (like glucosamine and chondroitin) showed statistically significant effects for pain relief in osteoarthritis, the quality of evidence was generally low, and the effect sizes were often too small to be considered clinically important. The only treatment showing a clinically important effect was the pharmaceutical anti-inflammatory drug, celecoxib.
· McAlindon TE, LaValley MP, Gulin JP, Felson DT. Glucosamine and chondroitin for treatment of osteoarthritis: a systematic quality assessment and meta-analysis. *JAMA. * 2000;283(11):1469-75.
· Conclusion: This early but influential systematic review found that when only high-quality, large trials were considered, the effect of glucosamine and chondroitin was minimal. It highlighted how study quality and publication bias could inflate estimates of treatment benefit.
These high-level evidence summaries highlight the critical gap between the plausible biological role of these nutrients and the demonstrable clinical benefits proven in rigorous trials. For reliable, updated information on specific supplements, the National Institutes of Health Office of Dietary Supplements (ods.od.nih.gov) and Harvard Health Publishing (www.health.harvard.edu) maintain excellent, evidence-based resources.









