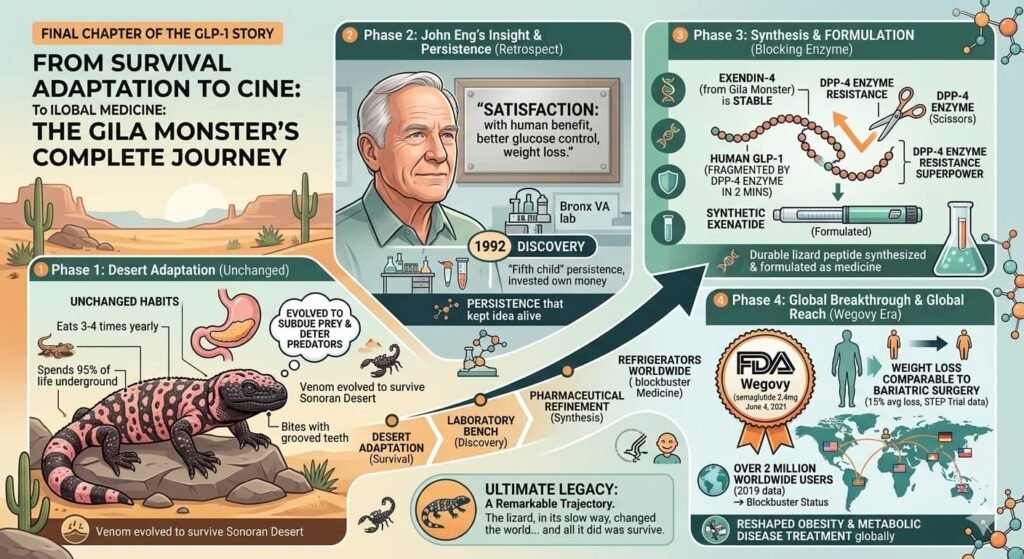
In the blistering Sonoran Desert, where temperatures regularly exceed 100 degrees Fahrenheit, a sluggish, beaded lizard emerges from its burrow perhaps four times a year to feed. The Gila monster (Heloderma suspectum) spends 95% of its life underground, surviving on massive seasonal feasts during which it consumes up to a third of its body weight in a single sitting. This extreme lifestyle—prolonged fasting punctuated by gluttony—required the evolution of a metabolic adaptation of genius-level sophistication: the ability to process enormous nutrient loads without crashing blood sugar systems.
What the Gila monster evolved over millions of years, a persistent New York endocrinologist named John Eng recognized in the 1980s as a potential key to unlocking one of medicine’s most persistent challenges: type 2 diabetes. The result of his insight would eventually reshape the treatment of metabolic disease worldwide, leading to a class of drugs that now includes Ozempic, Wegovy, Mounjaro, and Zepbound—medications that have been called the most important advance in metabolic health since insulin itself.
This is the story of how a desert lizard’s venom traveled from the rocks of Arizona to the pages of the New England Journal of Medicine, and how one scientist’s refusal to abandon an unfashionable idea changed the face of modern pharmacotherapy.
Part I: The Clue in the Saliva
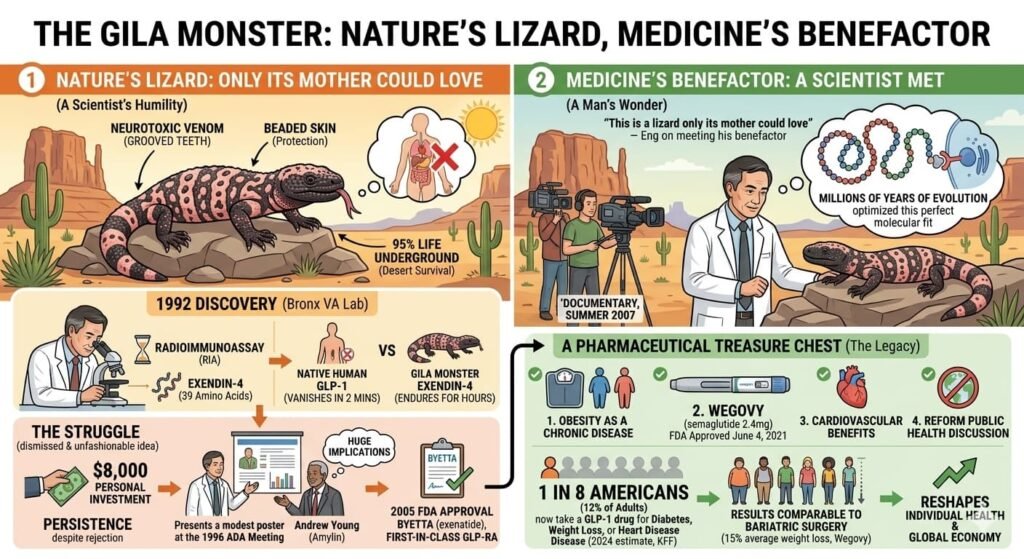
The Gila monster is not a cuddly creature. With its knobbly, black-and-pink beadwork skin, forked tongue, and neurotoxic venom delivered through grooved teeth rather than hollow fangs, it has inspired fear since the first settlers encountered it in the Gila River basin. The creature’s bite is painful—victims describe intense burning, swelling, and nausea—but rarely fatal to humans. What makes the Gila monster biologically extraordinary is not the venom’s toxicity but its metabolic purpose.
In the late 1980s, researchers began asking a deceptively simple question: How does an animal that eats so infrequently manage its blood sugar? When the Gila monster finally captures prey—a nest of rodent pups, perhaps, or bird eggs—its digestive system must process an enormous influx of nutrients. The lizard’s pancreas must secrete insulin in proportion to this massive glucose load. Yet between meals, sometimes for months, insulin requirements are minimal. The creature’s glucose-regulating system must therefore possess remarkable flexibility and, crucially, the hormonal signals that trigger insulin release must be extraordinarily durable.
Enter John Eng, a physician-researcher working at the James J. Peters Veterans Affairs Medical Center in the Bronx. Eng had trained under Rosalyn Yalow, who received the 1977 Nobel Prize in Physiology or Medicine for developing the radioimmunoassay technique—a method for measuring minute concentrations of hormones in blood. This background gave Eng both technical expertise and, perhaps more importantly, intellectual permission to pursue unconventional ideas.
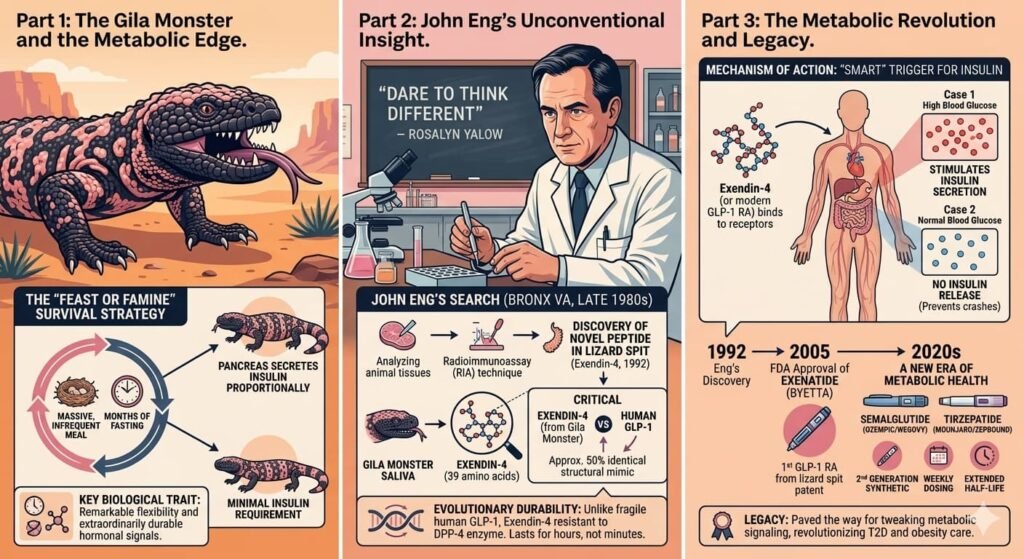
“She told me I should ‘dare to think different,'” Eng recalled of his mentor in a 2002 interview with the New York Times.
Eng began searching for new human hormones by analyzing animal tissues for chemical signals. His reasoning was elegant: if he could identify hormone-like compounds in animals with unusual metabolic adaptations, those molecules might reveal something about human physiology—or even become therapeutic agents themselves. He turned to the Gila monster because other scientists had observed that people bitten by the lizard sometimes developed pancreatitis, suggesting the venom contained something that stimulated the pancreas.
Using radioimmunoassay techniques, Eng analyzed Gila monster venom and saliva. He found something unexpected: two distinct peaks of hormonal activity. The smaller peak was expected—a known compound related to a hormone called glucagon. But the larger peak was something entirely new.
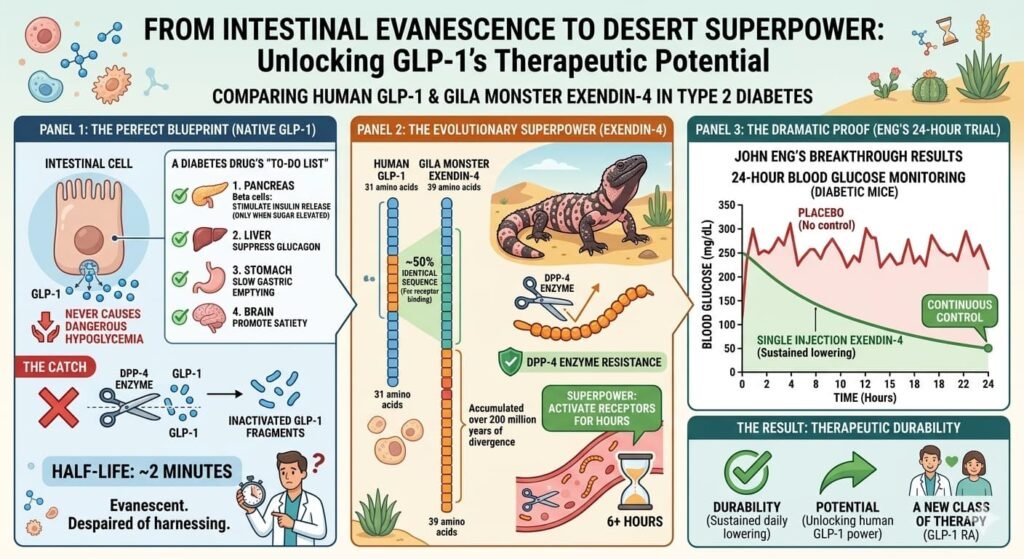
In 1992, Eng published his discovery of this novel peptide in the Journal of Biological Chemistry, naming it exendin-4. When he sequenced it, the result was striking: exendin-4 was approximately 50% identical to a human hormone called glucagon-like peptide-1 (GLP-1), which had recently been identified as a key regulator of blood sugar.
Part II: The Accidental Superpower
To understand why exendin-4 mattered, one must first understand GLP-1’s frustrating limitations.
GLP-1 is released from intestinal cells within minutes of eating. It travels to the pancreas and instructs beta cells to release insulin—but only when blood sugar is elevated, meaning it rarely causes dangerous hypoglycemia. It also suppresses glucagon (which raises blood sugar), slows gastric emptying (so nutrients enter the bloodstream gradually), and signals the brain to promote satiety. In short, GLP-1 does everything a diabetes drug should do.
There is, however, a catch: GLP-1 has a half-life in the human body of approximately two minutes. An enzyme called dipeptidyl peptidase-4 (DPP-4) cleaves and inactivates GLP-1 almost instantly, preventing it from reaching therapeutic targets in any meaningful way. For years, researchers had recognized GLP-1’s therapeutic potential but despaired of harnessing it. Native GLP-1 was simply too evanescent.
Exendin-4, by contrast, proved resistant to DPP-4 degradation. The structural differences between the lizard peptide and human GLP-1—differences accumulated over 200 million years of evolutionary divergence—conferred a superpower: exendin-4 could activate GLP-1 receptors for hours rather than minutes.
When Eng tested exendin-4 in diabetic mice, the results were dramatic. He measured blood glucose levels continuously for 24 hours and found that a single injection of exendin-4 produced sustained glucose lowering throughout the entire day. This was the durability researchers had dreamed of.
Part III: The Long Road to Recognition
Eng filed a patent application for exendin-4 on May 24, 1993. But here the story takes a dispiriting turn. His employer, the Department of Veterans Affairs, had no interest in commercializing the discovery. So Eng pursued pharmaceutical companies himself. For three years, he presented his findings at industry conferences and met with company representatives. The response was uniformly dismissive.
Jens Juul Holst, a Danish GLP-1 expert at the University of Copenhagen, later recalled Eng’s frustration to Science magazine in 2018: “He was extremely frustrated. Nobody was interested in his work. None of the important people. It was too strange for people to accept.”
The skepticism is understandable in context. The idea that a peptide from lizard venom could become a chronic therapy for a metabolic disease affecting millions seemed, on its face, preposterous. Moreover, peptides were considered poor drug candidates—they required injection, were expensive to manufacture, and were generally viewed as fragile molecules unsuitable for widespread therapeutic use.

Eng pressed on, investing approximately $8,000 of his own money to advance the work, according to a 2009 profile in the Beijing Evening News. He later described exendin-4 as “the fifth child of our family”—a creation he nurtured despite all evidence that the world was not interested.
The breakthrough came at the American Diabetes Association meeting in San Francisco in September 1996. Eng presented a modest poster summarizing his findings. Among those who stopped to examine it was Andrew Young, a physiologist at Amylin Pharmaceuticals, a small California biotechnology company focused on metabolic disease. Young immediately grasped the implications. But as he studied the poster, he noticed another scientist—a researcher from Eli Lilly—also taking careful notes.
What followed was a race. Young rushed back to Amylin and urged immediate action. Within weeks, Amylin pharmacologists confirmed exendin-4’s remarkable properties: it stimulated insulin secretion, suppressed glucagon, slowed gastric emptying, and reduced food intake. In October 1996, Eng licensed his patent to Amylin. By 2002, Amylin had partnered with Eli Lilly, which paid $325 million for development rights.
Part IV: From Venom to Pharmacy
The synthetic version of exendin-4, now called exenatide, required extensive pharmaceutical development. Scientists had to synthesize the 39-amino-acid peptide in quantities sufficient for clinical trials, formulate it for injection, and demonstrate safety and efficacy in thousands of patients.
The clinical trial program was extensive. Phase III studies enrolled more than 1,400 patients and demonstrated that exenatide effectively lowered blood glucose while producing an unexpected but welcome side effect: weight loss. Patients taking the drug lost an average of 2-3 kilograms, a striking contrast to many diabetes medications that promoted weight gain.
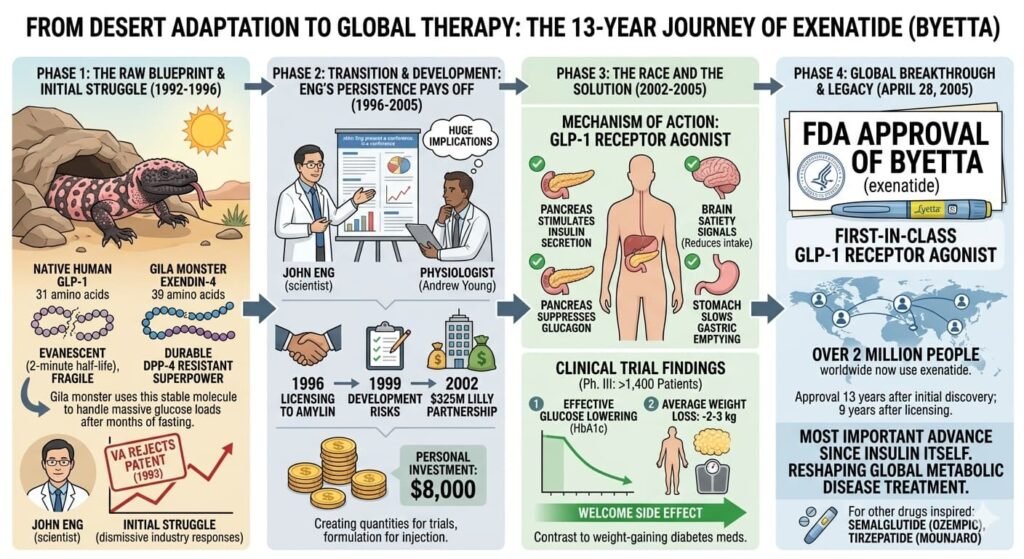
On April 28, 2005, the U.S. Food and Drug Administration approved Byetta (exenatide) for glycemic control in type 2 diabetes. It was the first GLP-1 receptor agonist to reach the market—a direct descendant of Eng’s lizard venom work. The approval came nearly 13 years after Eng’s initial discovery and nine years after he had finally licensed his patent.
The VA, which had initially declined to pursue the patent, later recognized the achievement. In a 2019 research summary, the agency noted: “More than 2 million people now use exenatide worldwide.”
Part V: The Evolution of an Idea
Byetta was a proof of concept, but it had limitations. Patients needed to inject it twice daily. Nausea was common. And while effective, its duration of action—while far longer than native GLP-1—was still measured in hours rather than days.
The pharmacological potential of GLP-1 receptor activation, however, was now established beyond question. Other companies entered the field. Novo Nordisk, a Danish diabetes specialist with decades of insulin expertise, began developing its own GLP-1 compounds. In 2010, the company received FDA approval for Victoza (liraglutide), a once-daily GLP-1 agonist.
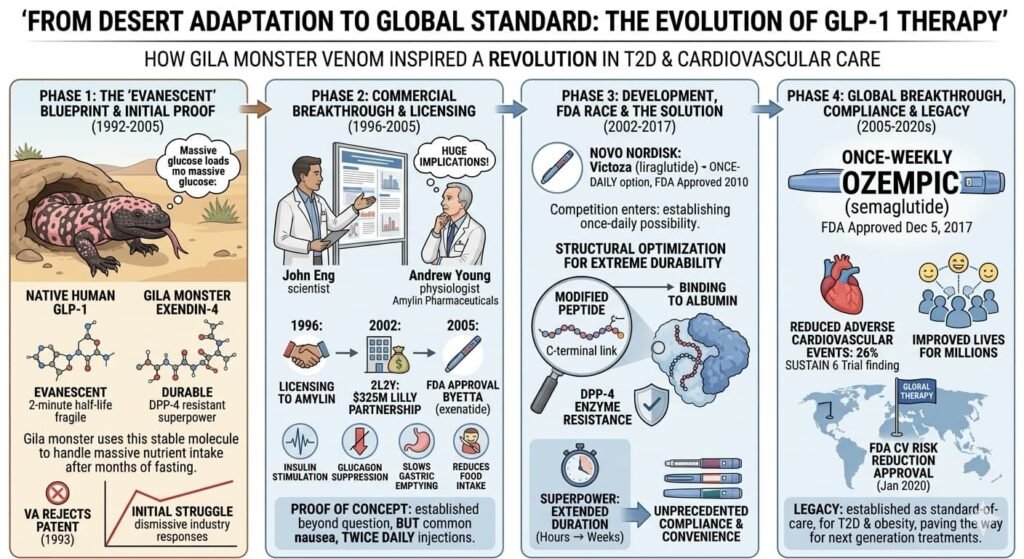
But the real prize was a once-weekly formulation that would transform convenience and compliance. Novo Nordisk achieved this with semaglutide, a molecule structurally optimized for extreme durability. By modifying the peptide to bind strongly to albumin (an abundant blood protein) and resist enzymatic degradation, scientists created a GLP-1 agonist that required injection only once weekly.
The SUSTAIN clinical trial program, encompassing multiple Phase III studies with thousands of patients, generated definitive efficacy and safety data. On December 5, 2017, the FDA approved Ozempic (semaglutide) for type 2 diabetes. Subsequent trials demonstrated cardiovascular benefits: in the SUSTAIN 6 trial, semaglutide reduced major adverse cardiovascular events by 26% in high-risk patients, a finding that led to FDA approval for cardiovascular risk reduction in January 2020.
Part VI: The Weight-Loss Revolution
Throughout the GLP-1 development story, one observation recurred with striking consistency: patients lost weight. This was not merely a cosmetic side effect but a potentially therapeutic benefit, given that obesity underlies much of type 2 diabetes.
Novo Nordisk pursued this indication systematically. Higher doses of semaglutide were tested specifically for weight management. The STEP clinical trial program demonstrated that semaglutide 2.4 mg produced average weight loss of approximately 15% of body weight—results comparable to bariatric surgery.
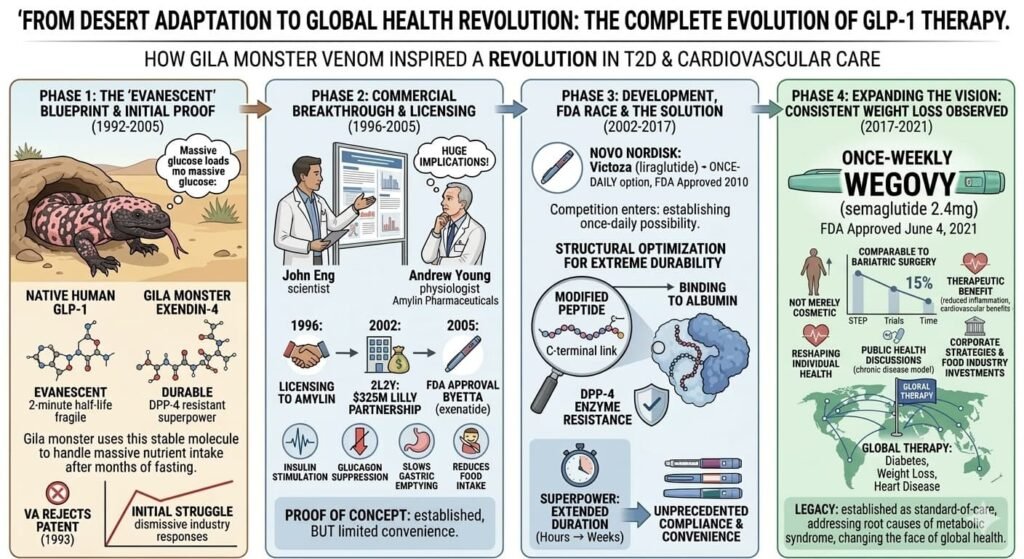
On June 4, 2021, the FDA approved Wegovy (semaglutide) for chronic weight management in adults with obesity or overweight with at least one weight-related condition. The approval marked a turning point: a drug derived from Gila monster venom had become a mainstream treatment for a condition affecting more than 40% of American adults.
The impact has been extraordinary. By 2024, an estimated 12% of American adults reported taking a GLP-1 medication for diabetes, weight loss, or heart disease, according to data from the Kaiser Family Foundation. The drugs have been credited with reshaping not only individual health but also corporate strategies, food industry investments, and public health discussions about obesity as a chronic disease requiring medical treatment.
Part VII: The Broader Canvas of Venom Therapeutics
The Gila monster’s contribution to medicine is not an isolated curiosity. Animal venoms have proven to be among nature’s most sophisticated pharmacological libraries—molecular arsenals honed by evolution for predation and defense that often target precisely the same receptors and channels that interest drug developers.
The classic precedent is captopril, the first ACE inhibitor for hypertension, developed from Brazilian pit viper (Bothrops jararaca) venom. In the 1960s, Brazilian scientist Sérgio Ferreira discovered that the viper’s venom contained peptides that potentiated bradykinin, a vasodilator. These bradykinin-potentiating factors ultimately led to captopril’s development, transforming cardiovascular medicine.
Another striking example is ziconotide (Prialt), an analgesic derived from the venom of the cone snail Conus magus. This peptide selectively blocks N-type calcium channels in the spinal cord, providing pain relief for severe chronic pain without the tolerance and addiction risks of opioids. It was approved by the FDA in 2004.
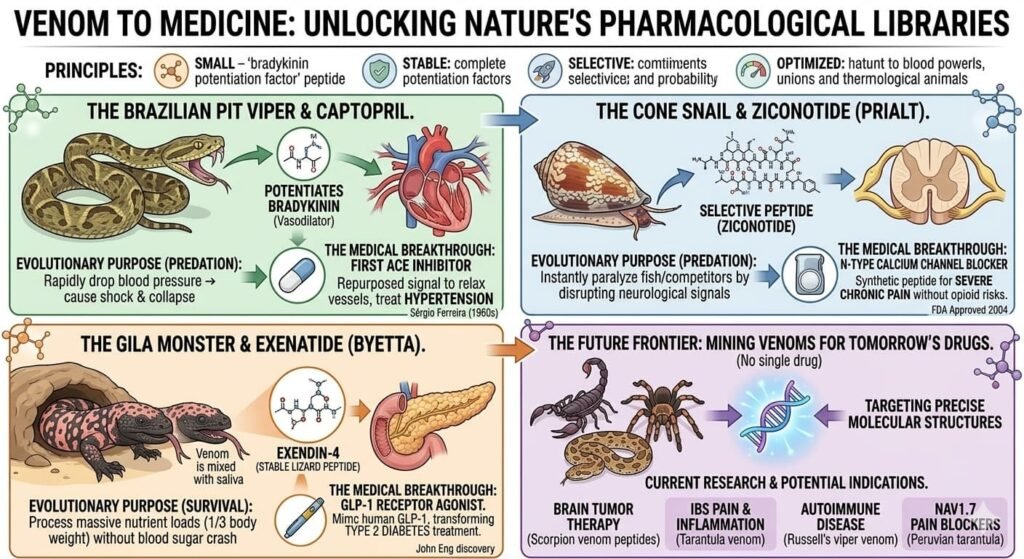
The list continues to grow. Scorpion venom has yielded peptides being investigated for brain tumor therapy. Tarantula venom contains compounds that may treat irritable bowel syndrome pain. Russell’s viper venom has inspired molecules for autoimmune disease. The Peruvian tarantula (Avicularia juruensis) produces a peptide that inhibits Nav1.7, a sodium channel implicated in pain signaling, offering hope for new analgesics.
These successes reflect fundamental principles: venom peptides are typically small, stable, and exquisitely selective for their molecular targets. They have been optimized by millions of years of evolution to disrupt specific physiological processes—precisely the characteristics sought in drug development.
Part VIII: Meeting the Monster
In the summer of 2007, a television crew making a documentary about the discovery of exendin-4 took John Eng to the Arizona desert. There, for the first time, the New York endocrinologist came face to face with the creature that had made his scientific reputation.
The Gila monster, placid and unimpressed by the commotion, allowed itself to be observed. Eng studied the animal with the curiosity of a scientist and, perhaps, the wonder of a man meeting a benefactor.
“This is a lizard only its mother could love,” he told reporters from the Montreal Gazette in a 2023 interview reflecting on the encounter.
The observation was characteristically self-deprecating, but it missed something essential. The Gila monster, with its beaded skin, sluggish movements, and reclusive habits, had proven to be a pharmaceutical treasure chest. Its venom, evolved to subdue prey and deter predators in the harsh Sonoran Desert, contained a molecule that spoke to human metabolic receptors as though designed for the purpose.
Part IX: The Ongoing Story
The Gila monster’s contribution to medicine continues to evolve. New GLP-1 drugs have emerged, including Eli Lilly’s tirzepatide (Mounjaro for diabetes, Zepbound for obesity), which activates both GLP-1 and GIP receptors and has produced even greater weight loss than semaglutide. Oral formulations—Rybelsus for semaglutide—have freed patients from injections.
Research continues into additional applications. GLP-1 drugs appear to have benefits beyond glucose and weight: reduced cardiovascular events, potential kidney protection, and even possible effects on neurodegenerative conditions and addiction. The FLOW trial, a double-blind, placebo-controlled study examining renal outcomes in patients with type 2 diabetes and chronic kidney disease, demonstrated that semaglutide reduced the progression of kidney disease by 24% compared to placebo. These results, published in the New England Journal of Medicine in 2024, exemplify the expanding therapeutic horizons of this drug class.
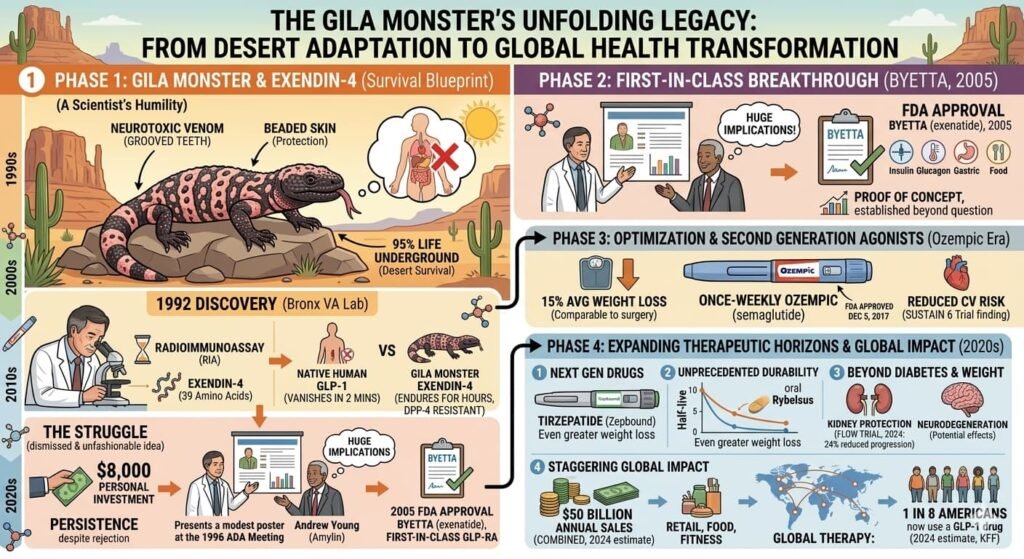
The economic impact has been staggering. By 2024, GLP-1 drugs had become blockbusters, with combined sales approaching $50 billion annually. The drugs have reshaped entire sectors: retail, food, and fitness industries all anticipate changed consumer behavior as obesity becomes more treatable.
Yet the fundamental insight remains John Eng’s: that a desert lizard’s survival strategy could illuminate a path to treating human metabolic disease. When Eng filed his patent in 1993, GLP-1 was a scientific curiosity with no therapeutic future. The conventional wisdom held that peptide hormones could never become practical drugs—they were too unstable, too expensive, too difficult. Eng’s persistence, nurtured by his training under a Nobel laureate who had herself challenged orthodoxy, kept the idea alive long enough for science to catch up.
Part X: Lessons for Drug Discovery
The Gila monster story offers several enduring lessons for pharmaceutical development.
First, nature remains the most creative chemist. Despite advances in computational drug design and high-throughput screening, evolution has produced molecular solutions to physiological problems that human inventors have not yet imagined. Venoms, in particular, represent an unexplored pharmacopeia. The number of venomous species is estimated at more than 200,000, yet only a tiny fraction have been studied for drug potential.
Second, unconventional sources deserve attention. The idea that a desert lizard’s venom could treat a chronic metabolic disease seemed fanciful when Eng first proposed it. The scientific community’s skepticism was predictable but wrong. The history of medicine is replete with such examples: penicillin from mold, cyclosporine from soil fungus, statins from red yeast rice. The next breakthrough may come from an equally unlikely source.
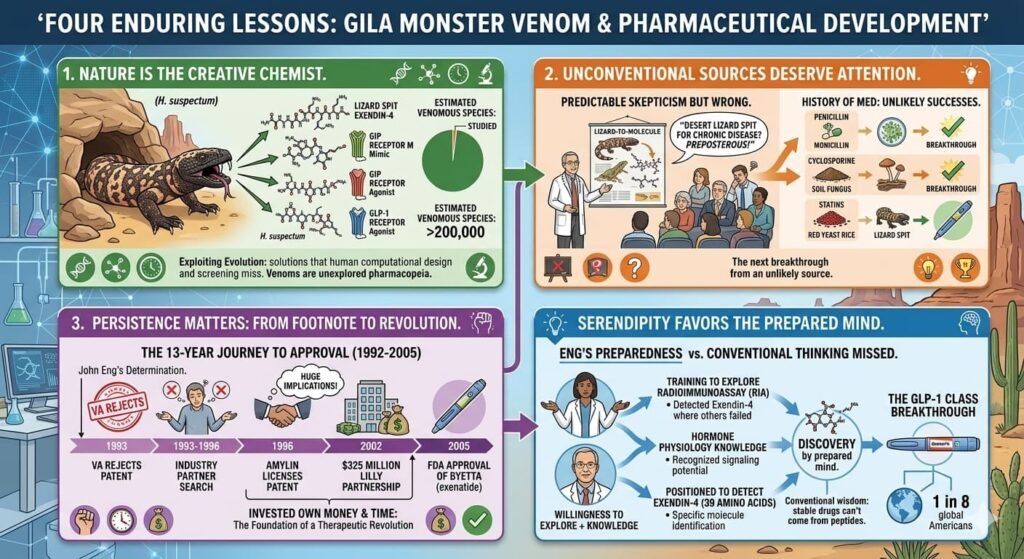
Third, persistence matters. Eng spent three years seeking industry partners before Amylin finally licensed his patent. He invested his own money and time in a project that most experts dismissed. Without his determination, exendin-4 might have remained a footnote in the toxinology literature rather than becoming the foundation of a therapeutic revolution.
Fourth, serendipity favors the prepared mind. Eng’s training in radioimmunoassay gave him tools to detect exendin-4 that were not available to most researchers. His knowledge of hormone physiology allowed him to recognize the peptide’s significance. And his willingness to explore an unusual biological system—lizard venom—positioned him to make a discovery that more conventional researchers missed.
Epilogue: The Lizard’s Legacy
The Gila monster still lives in the Sonoran Desert, its habits unchanged by the medical revolution it inadvertently enabled. It still eats three or four times yearly, still spends most of its life underground, still bites with grooved teeth when threatened. The creature’s venom, which evolved to incapacitate small prey and discourage predators, now circulates in millions of human patients in the form of synthetic exenatide and its descendants.
John Eng, now retired, occasionally reflects on the journey. In interviews, he has expressed satisfaction not only with the scientific achievement but with the human benefit. Millions of patients with diabetes have achieved better glucose control. Millions more with obesity have lost weight and improved their health. The lizard’s venom, refined and synthesized and formulated, has become medicine.
It is a remarkable trajectory: from the desert floor to the laboratory bench, from the venom gland of a reclusive reptile to the refrigerators of patients worldwide, from scientific obscurity to blockbuster status. The Gila monster, in its slow, patient way, has changed the world.
And all it did was survive.
All Images are AI Generated for Illustration Only. E&OE
The author was formerly executive director of Indian Journal of Clinical Practice in an era when there were only JAMA and BMJ as the leading medical journals available for Indian clinicians. Indian Journal of Clinical Practice became the pioneer in medical publishing providing customised scientific inputs to over 20 therapy areas from the early 1990s.
Appendix: Sources and Further Reading
Primary Scientific Publications
- Eng J, Kleinman WA, Singh L, Singh G, Raufman JP. “Isolation and characterization of exendin-4, an exendin-3 analogue, from Heloderma suspectum venom.” Journal of Biological Chemistry. 1992;267(11):7402-7405.
- Raufman JP. “Bioactive peptides from lizard venoms.” Regulatory Peptides. 1996 Jan 16;61(1):1-18.
- Drucker DJ, Nauck MA. “The incretin system: glucagon-like peptide-1 receptor agonists and dipeptidyl peptidase-4 inhibitors in type 2 diabetes.” The Lancet. 2006 Nov 11;368(9548):1696-705.
- Marso SP, Bain SC, Consoli A, et al. “Semaglutide and Cardiovascular Outcomes in Patients with Type 2 Diabetes.” New England Journal of Medicine. 2016 Nov 10;375(19):1834-1844. (SUSTAIN-6)
- Perkovic V, Tuttle KR, Rossing P, et al. “Effects of Semaglutide on Chronic Kidney Disease in Patients with Type 2 Diabetes.” New England Journal of Medicine. 2024;391:109-121. (FLOW trial)
- Wilding JPH, Batterham RL, Calanna S, et al. “Once-Weekly Semaglutide in Adults with Overweight or Obesity.” New England Journal of Medicine. 2021 Mar 18;384(11):989-1002. (STEP 1)

Regulatory Documents and Official Sources
- U.S. Food and Drug Administration. “Drug Approval Package: Byetta (Exenatide) Injection.” Application No.: 021773. Approval Date: April 28, 2005. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/nda/2005/021773_byettatoc.cfm
- U.S. Food and Drug Administration. “FDA approves Ozempic (semaglutide) for type 2 diabetes.” December 5, 2017. Available at: https://www.fda.gov/news-events/press-announcements/fda-approves-new-drug-treatment-type-2-diabetes
- U.S. Food and Drug Administration. “FDA approves Wegovy (semaglutide) for chronic weight management.” June 4, 2021. Available at: https://www.fda.gov/news-events/press-announcements/fda-approves-new-drug-treatment-chronic-weight-management-first-2014
- U.S. Department of Veterans Affairs. “Diabetes drug from Gila monster venom.” VA Research in Action. May 7, 2019. Available at: https://www.research.va.gov/research_in_action/Diabetes-drug-from-Gila-monster-venom.cfm
- European Medicines Agency. “Ozempic: Summary of product characteristics and regulatory history.” Available at: https://www.ema.europa.eu/en/medicines/human/EPAR/ozempic

News Media and Interviews
- Kolata G. “Lizard-linked therapy has roots in the Bronx.” New York Times. September 21, 2002. [Contains Eng’s quote about Yalow]
- Winkler S. “How a Lizard’s Venom Gave Us a New Class of Diabetes Drugs.” Science magazine. 2018. [Contains Holst’s quote about Eng’s frustration]
- 北京晚报 [Beijing Evening News]. “希拉巨蜥的故事” [The Story of the Gila Monster]. August 28, 2009. Available at: http://news.sina.com.cn/c/2009-08-28/151016202029s.shtml [Source for Eng’s $8,000 personal investment]
- Schwarcz J. “The Right Chemistry: How Gila monster venom led to a blockbuster drug.” Montreal Gazette. 2023. [Contains Eng’s “lizard only its mother could love” quote]
- Molteni M. “The Lizard Venom That Became a Blockbuster Weight-Loss Drug.” Wired. 2023.
- Le Quotidien du Pharmacien. “Le trésor caché du monstre de Gila” [The Hidden Treasure of the Gila Monster]. March 9, 2017. Available at: https://prod.lequotidiendupharmacien.fr/formation/specialites-medicales/le-tresor-cache-du-monstre-de-gila

Review Articles and Broader Context
- Kaiser Family Foundation. “KFF Health Tracking Poll: The public’s views on new prescription weight-loss drugs.” October 2024. Available at: https://www.kff.org/health-costs/poll-finding/kff-health-tracking-poll-views-on-weight-loss-drugs/ [Source for 12% figure]
- BioSpace. “Deadly Venom is Being Turned into Life-Saving Medical Treatments.” May 16, 2022. Available at: https://www.biospace.com/article/deadly-venom-being-turned-into-life-saving-medical-treatments/
- UT Southwestern Medical Center. “A powerhouse of obesity research – then and now.” September 20, 2024. Available at: https://utswhealth.net/newsroom/articles/year-2024/sept-obesity-research.html
- NIH National Library of Medicine. “Animal Venom in Modern Medicine: A Review of Therapeutic Applications.” Toxins. 2025 Jul 28;17(8):371. doi: 10.3390/toxins17080371. Available at: https://pmc.ncbi.nlm.nih.gov/articles/PMC12390438/
Key Patent Information
- Eng J. “Exendin and exendin analogs.” U.S. Patent 5,424,286. Filed May 24, 1993; Issued June 13, 1995. (First patent filing for exendin-4)
- Novo Nordisk. “GLP-1 derivatives.” U.S. Patent 6,268,343. Filed January 28, 1999. (Liraglutide)
- Novo Nordisk. “Derivatives of GLP-1 analogs.” U.S. Patent 8,129,343. Filed January 2, 2002. (Semaglutide)