For decades, the Medical Representative has been the backbone of the pharmaceutical industry.
No product launch, brand strategy, or market expansion truly succeeded without the relentless field presence of MRs.
Yet today, across the industry, there is an uncomfortable silence around a growing problem. The MR model is under stress. Everyone senses it. Few openly discuss it.

The Changing Terrain: The environment in which MRs operate today is radically different from what it was even 10–15 years ago.
Doctors are busier than ever.
Hospital systems and corporate chains increasingly control access.
Digital channels compete for physician attention.
Compliance rules restrict traditional promotional practices.
The result is a paradox: Expectations from MRs have increased, while access and influence have decreased.

The Productivity Pressure: Pharma companies continue to rely heavily on field forces.
But many MRs are now caught in an uncomfortable squeeze:
• Higher call targets
• Expanding product portfolios
• More demanding reporting systems
• Reduced doctor interaction time
The industry has responded with dashboards, CRM tools, digital aids, and analytics. Yet the question remains: Are we solving the problem—or simply measuring it better?

The Silent Morale Crisis – Another issue rarely discussed openly is MR morale.
Across many companies, field teams report:
• Increasing administrative workload
• Unrealistic performance targets
• Limited career progression
• Constant pressure with little recognition
This does not mean the MR profession is disappearing. But it does mean the traditional MR model is being stretched beyond its limits.

The Capability Gap – The role of the MR is also evolving.
Today’s successful representatives must combine:
• Scientific understanding
• Digital engagement skills
• Business intelligence
• Relationship management
However, training systems across the industry have not always kept pace with these expectations. Many MRs are being asked to perform 21st-century roles with 20th-century training models.

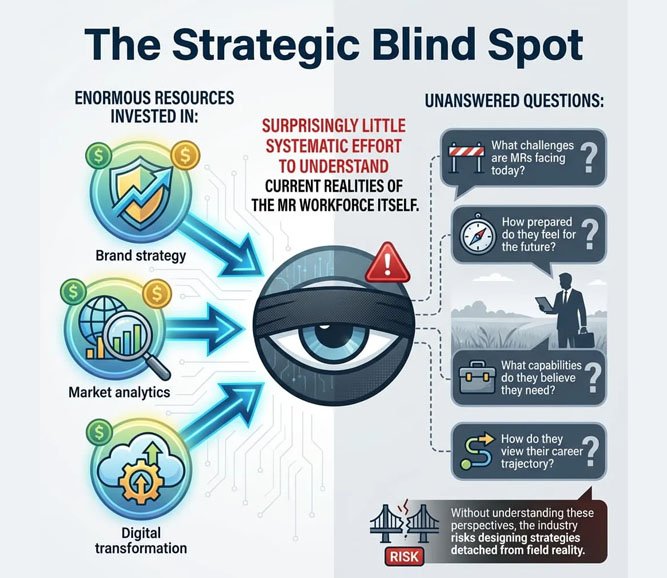
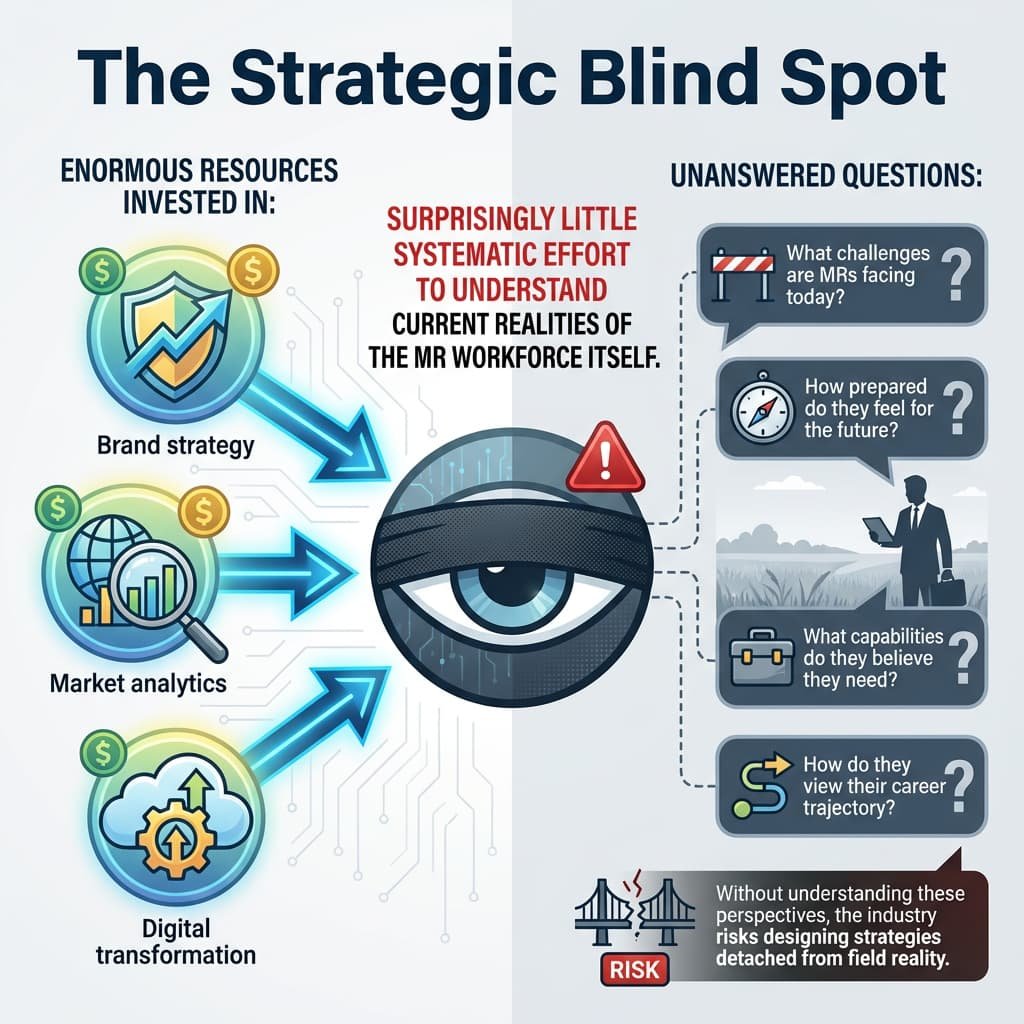
The Strategic Blind Spot
Pharma companies invest enormous resources in:
• Brand strategy
• Market analytics
• Digital transformation
But surprisingly little systematic effort has been made to understand the current realities of the MR workforce itself.
Questions remain largely unanswered:
• What challenges are MRs facing today?
• How prepared do they feel for the future?
• What capabilities do they believe they need?
• How do they view their career trajectory?
Without understanding these perspectives, the industry risks designing strategies detached from field reality.
Why This Conversation Matters? The MR remains one of the most powerful connectors between pharmaceutical science and clinical practice. But the profession is at an inflection point.
Ignoring the structural pressures surrounding the MR role will only lead to:
• declining motivation
• rising attrition
• reduced field effectiveness
On the other hand, confronting these issues openly could unlock a new generation of future-ready MRs.

A Step Toward Understanding – To bring clarity to this issue, MedicinMan, in partnership with SlamDunk, has launched the Future-Ready Super MR: Industry Capability, Engagement & Career Outlook Survey 2026.
The goal is simple: to listen to the voices of Medical Representatives themselves.
If you are a Medical Representative—or work closely with field teams—your insights will help build one of the most meaningful industry snapshots of the MR profession today.
The survey takes about 10 minutes.
Everyone who completes the survey will receive the final report free.
Take the survey here:
https://forms.gle/wfg5YucnYc64qk4x9
Your participation will help the industry better understand the real challenges, aspirations, and future of Medical Representatives.