In September 2011, MedicinMan published a UCPMP Special Issue titled: “UCPMP: Does Healthcare Need a Anna Hazare?” At a time when India’s anti-corruption movement dominated national discourse, we asked whether a voluntary marketing code would be enough to reform pharmaceutical promotion practices.
Fifteen years later, that question remains structurally relevant.
Recent disclosures before the Madras High Court — including statements by the Income Tax Department of India regarding promotional expense classifications — suggest that marketing governance and tax treatment remain deeply intertwined. This article traces the evolution of UCPMP from its early voluntary phase to today’s urgent call for statutory transparency.
I. 2011 — The Foundational Question In its September 2011 issue (Vol. 1, Issue 2), MedicinMan:
• Identified UCPMP as a structural intervention by the Department of Pharmaceuticals.
• Framed pharmaceutical marketing ethics within the broader anti-corruption climate.
• Questioned whether voluntary compliance could reshape entrenched commercial practices.
At that time:
• UCPMP was advisory.
• Enforcement mechanisms were limited.
• Disclosure infrastructure did not exist.
• Compliance depended largely on self-regulation.
Our editorial stance was clear: Codes without enforceability risk becoming symbolic.

II. 2011–2022: Incremental Reform, Structural Gaps
Over the next decade:
• UCPMP iterations were issued and revised.
• Industry associations were expected to handle grievances.
• Public disclosure remained absent.
• Marketing practices continued under evolving interpretation.
The inflection point came with the Supreme Court ruling in CIT v. Apex Laboratories Pvt. Ltd. (2022), which held that freebies given in violation of medical ethics regulations are not deductible under Section 37(1) of the Income Tax Act. That judgment aligned tax law with ethical norms. But alignment through litigation is reactive, not systemic.

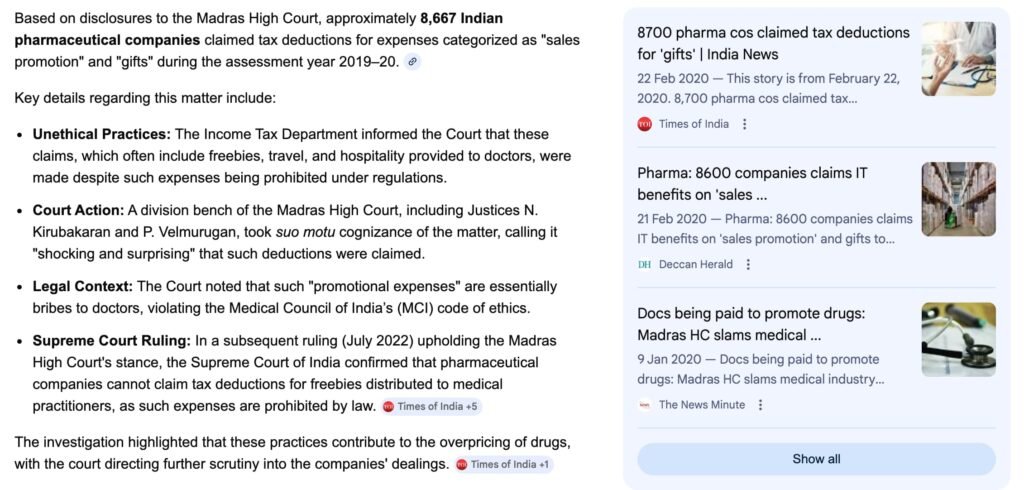
III. The 8,700 Moment — A Governance Stress Test
Disclosures before the Madras High Court indicated that approximately 8,700 pharmaceutical companies claimed deductions under “sales promotion” and “gifts” in FY 2019–20. Whether each claim was improper is a matter of scrutiny.
But the scale suggests:
• Widespread classification practices.
• Persistent ambiguity between marketing expenditure and ethical compliance.
• A need for integrated regulatory architecture.
This is no longer about individual misconduct. It is about systemic design.
IV. The Structural Gap
India’s current framework involves:
• Marketing code oversight — Department of Pharmaceuticals
• Medical ethics regulation — National Medical Commission framework
• Price control enforcement — National Pharmaceutical Pricing Authority
• Tax deductibility — Income Tax authorities
These operate in silos. Without transparency infrastructure, enforcement becomes episodic rather than preventive.


V. Global Benchmark: Transparency as Enforcement
The U.S. Physician Payments Sunshine Act requires mandatory disclosure of payments to physicians, overseen by the Centers for Medicare & Medicaid Services through the Open Payments database.
Public disclosure:
• Enables scrutiny
• Reduces ambiguity
• Protects compliant firms
• Aligns governance with capital market expectations
Transparency becomes a compliance multiplier.

VI. Reform Roadmap: From Voluntary Code to Statutory Transparency
Phase 1: Codify UCPMP into Binding Law
• Remove interpretive ambiguity.
• Establish clear penalties.
• Define prohibited transfers precisely.
Phase 2: National Disclosure Portal
Mandatory quarterly reporting of:
• Company name
• Healthcare professional name
• Amount
• Nature of transfer
• Purpose
Publicly searchable.
Phase 3: Tax Synchronization
• Deny deduction for undisclosed transfers.
• Integrate portal data with tax filings.
• Mandatory statutory auditor certification.
Phase 4: Accountability Escalation
• Financial penalties
• Public naming
• Tender disqualification for repeat violations

VII. Why 2011 Still Matters
In 2011, we asked whether voluntary reform would be sufficient.
In 2026, the answer is clearer: Voluntary codes start conversations.
Statutory transparency builds systems.
The Indian pharmaceutical industry is entering a phase defined by:
• Innovation
• Biologics
• Global partnerships
• Institutional capital
• ESG scrutiny
Governance maturity must scale alongside commercial ambition.

Conclusion
The journey from 2011 to today reveals a pattern: Intent → Guidance → Judicial Correction → Structural Pressure The next logical step is institutional transparency. Not as punishment. But as protection — for ethical firms, for investors, and for patients.
Fifteen years ago, MedicinMan raised the question. Today, the industry must deliver the answer.

All Images are AI Generated for Illustration Only. E&OE
References & Source Documentation
- Uniform Code of Pharmaceutical Marketing Practices (UCPMP)
Department of Pharmaceuticals (DoP), Government of India.
Uniform Code of Pharmaceutical Marketing Practices (UCPMP) – Various editions (2011 onwards).
Issued by the Department of Pharmaceuticals, Ministry of Chemicals & Fertilizers, Government of India.
• Initial voluntary framework introduced circa 2011–2014.
• Subsequent revisions strengthened compliance expectations.
• Latest iterations move toward enforceable mechanisms.
Official notifications available via the DoP website.
- Income Tax Treatment of Promotional Expenditure
Central Board of Direct Taxes (CBDT).
Circular No. 5/2012 dated 1 August 2012.
Clarified that expenses incurred in violation of regulations (including medical council guidelines) are not allowable under Section 37(1) of the Income Tax Act, 1961.
Issued by the Central Board of Direct Taxes.
- Supreme Court Judgment
Commissioner of Income Tax v. Apex Laboratories Pvt. Ltd. (2022)
Supreme Court of India.
The Supreme Court of India held that freebies provided to medical practitioners in violation of the Indian Medical Council Regulations are not deductible business expenses under Section 37(1).
This judgment established alignment between medical ethics regulations and tax deductibility.
- Madras High Court Proceedings (8,700 Companies Disclosure Context)
Proceedings before the Madras High Court referencing submissions from the Income Tax Department regarding approximately 8,700 pharmaceutical companies claiming deductions under promotional expense classifications (FY 2019–20).
Court filings and related coverage available via public judicial records and national financial press.
- National Pharmaceutical Pricing Authority (NPPA)
The National Pharmaceutical Pricing Authority operates under the Ministry of Chemicals & Fertilizers and enforces the Drugs (Prices Control) Order (DPCO).
Provides contextual relevance for regulatory fragmentation discussed in the white paper.
- U.S. Sunshine Act – Global Transparency Benchmark
Physician Payments Sunshine Act (Section 6002 of the Affordable Care Act, 2010).
Administered by the Centers for Medicare & Medicaid Services through the Open Payments database.
Mandates public disclosure of payments or transfers of value from pharmaceutical and medical device manufacturers to physicians and teaching hospitals.
Public database: Open Payments (CMS).
- MedicinMan Archival Reference
MedicinMan.
Vol. 1, Issue 2 (September 2011).
“UCPMP: Does Healthcare Need a Anna Hazare?”
UCPMP Special Issue.
Archival copy available in MedicinMan’s print and SlideShare repository.
Here are the direct links to the MedicinMan articles on UCPMP referenced earlier. These are from the official site and archives.
Primary Links
- National Seminar on UCPMP: https://medicinman.net/2016/08/national-seminar-on-uniform-code-of-pharmaceutical-marketing-practices-ucpmp/[1]
- Tech Advances Making UCPMP Obsolete?: https://medicinman.weslamdunk.com/2020/10/have-tech-advances-made-ucpmp-obsolete-need-for-contemporary-pharma-marketing-code/[2]
- Five Disruptive Forces (incl. UCPMP): https://medicinman.net/2016/07/five-disruptive-forces-shaking-up-indian-pharma/[3]
Site Archives
- MedicinMan Homepage: https://medicinman.net[4]
- Special Announcements Category: https://medicinman.net/category/special-announcement/[5]
Sources
[1] National Seminar on UCPMP | MedicinMan https://medicinman.net/2016/08/national-seminar-on-uniform-code-of-pharmaceutical-marketing-practices-ucpmp/
[2] Have Tech Advances Made UCPMP Obsolete? Need … – MedicinMan https://medicinman.weslamdunk.com/2020/10/have-tech-advances-made-ucpmp-obsolete-need-for-contemporary-pharma-marketing-code/
[3] Five Disruptive Forces Shaking Up Indian Pharma | MedicinMan https://medicinman.net/2016/07/five-disruptive-forces-shaking-up-indian-pharma/
[4] MedicinMan | Amplifying Healthcare Voices https://medicinman.net
[5] Special Announcement – MedicinMan https://medicinman.net/category/special-announcement/